While chemotherapy medications are beneficial in the treatment of cancer, they can have unwanted adverse effects on many organs, including the heart. Cardiotoxicity is the term used to describe the damage that certain chemotherapy medicines cause to the heart and its muscles. This can result in illnesses such as cardiomyopathy (heart muscle weakness) or heart failure. The University of Illinois at Chicago researchers discovered a method for enzymes to assist avoid cardiac damage in chemotherapy patients.
The enzymes are generally found in a cell’s mitochondria, the powerhouse that generates energy. When cardiac cells are stressed by specific types of chemotherapeutic treatments, the enzymes go into the cell’s nucleus, where they can keep the cells alive. The paper was published in Nature Communications.
“As chemotherapy has become more effective, we are seeing an increase in the number of cancer survivors.” But the terrible side is that many of these survivors are now suffering from heart failure,” said co-senior author Sang Ging Ong, an assistant professor of pharmacology and medicine.
Assessing the injury caused by chemotherapy and the enzyme movement from the mitochondria into the nucleus of those heart cells in a lab would help determine what the patient’s likely response would be to chemotherapy.
Dr. Jalees Rehman
As a result, a new field known as cardio-oncology has emerged. The majority of earlier research in the field concentrated on the methods through which chemotherapeutic medicines harmed the mitochondria of cardiac cells. This research team was curious about a different angle: Why do some patients’ hearts escape damage? Is there something specific about their cells that protects them?
First, the researchers noticed that when heart cells were stressed by chemotherapy, mitochondrial enzymes went into the cell’s nucleus, which was an unusual occurrence. Dr. Jalees Rehman, co-senior author, Benjamin Goldberg Professor and director of the UIC Department of Biochemistry and Molecular Genetics, noted that they didn’t know if the movement was the source of the cell’s damage or a means of defence.
“We really didn’t know which way it would go,” he said.
To find out, the researchers generated versions of the enzymes that would specifically move into the nucleus and bypass the mitochondria. They discovered that this relocation fortified the cells, keeping them alive. They demonstrated that this process worked in both heart cells generated from human stem cells and in mice exposed to chemotherapy.
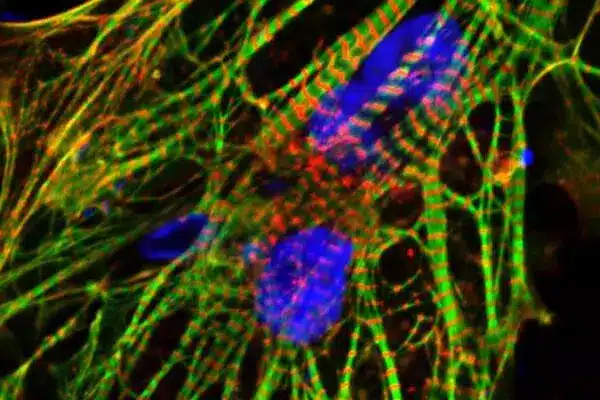
“This appears to be a new mechanism by which heart cells can defend themselves against chemotherapy damage,” said Rehman, who works at the University of Illinois Cancer Center.
The discovery raises the prospect of novel clinical applications. Individual patients might be tested to see if heart cells created from customized stem cells will effectively protect themselves from chemotherapy by relocating enzymes from the mitochondria to the cell’s nucleus. Doctors might take blood from the patient, create stem cells from the blood cells, and then use those individualized stem cells to create heart cells that have the same genetic makeup as the patient’s heart cells.
“Assessing the injury caused by chemotherapy and the enzyme movement from the mitochondria into the nucleus of those heart cells in a lab would help determine what the patient’s likely response would be to chemotherapy,” Rehman added.
In those with insufficient protection, one could improve it by enhancing enzyme mobility and heart cell protection.
More research is needed to see whether this technique can help prevent heart damage caused by other disorders such as high blood pressure and heart attacks, as well as whether it works in other cells such as those in blood vessels.