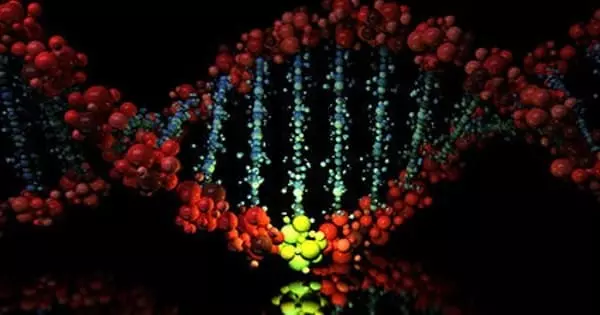
Genome editing is a technique for modifying specific sections of a genome. Scientists have been able to alter DNA since the 1970s, but in recent years, they have developed faster, less expensive, and more precise methods of adding, removing, or changing genes in living organisms. Researchers are working to create therapies that use gene editing to treat children and adults with a variety of conditions such as sickle cell disease, hemophilia, and some forms of cancer and blindness. Researchers have devised a method for precisely and quickly correcting genetic changes in cultured patient cells.
The University of Helsinki and University Hospital Helsinki researchers have developed a method for precisely and rapidly correcting genetic alterations in cultured patient cells. From a 2-3 mm skin biopsy taken from patients with various genetic diseases, the method generates genetically corrected autologous pluripotent stem cells. The corrected stem cells are essential in the research and for the development of new therapies for the diseases in question.
The new method is based on previous groundbreaking research in the fields of stem cells and gene editing, including two Nobel Prize-winning techniques. The first technique is the discovery of induced pluripotent stem cells, or iPSCs, which won the Nobel Prize in 2012. The CRISPR-Cas9 “gene scissors” innovation, which won the prize in 2020, is the other technique. The new method combines these techniques to correct gene changes that cause inherited diseases while also producing fully functional new stem cells.
Our new system is much faster and more precise than the older methods in correcting DNA errors, and the speed makes it easier and reduces the risk of unwanted changes.
Professor Kirmo Wartiovaara
New therapeutic possibilities to inherited diseases
The researchers’ long-term goal is to create autologous cells with therapeutic properties. The use of the patient’s own corrected cells may aid in avoiding the immunological challenges that impede donor organ and tissue transplantation. The new method was developed by Sami Jalil, an Argentine Ph.D. student at the Biomedicum Helsinki Stem Cell Center, and it was recently published in Stem Cell Reports, the journal of the International Society of Stem Cell Research.
There are over 6000 known inherited diseases that are caused by various gene alterations. Some are currently receiving treatment with a cell or organ transplant from a healthy donor if one is available.

“Our new system is much faster and more precise than the older methods in correcting DNA errors, and the speed makes it easier and reduces the risk of unwanted changes,” says adjunct professor Kirmo Wartiovaara, who oversaw the research.
“We have achieved up to 100 percent efficacy under ideal conditions, but it is important to remember that the correction of cultured cells is still a long way from proven therapeutic applications. However, it is a very promising start “Wartiovaara continues.
New technologies frequently raise ethical concerns about unknown risks and benefits. These questions become especially difficult and critical when we discuss human germline editing, which affects future generations who, obviously, cannot consent to the changes being made to their DNA.
What risks would women (who are rarely mentioned in discussions about human gene editing for reproduction) face as the ones carrying pregnancies started with genetically modified embryos and delivering the resulting children (for themselves or for others) face? How could prospective parents make informed decisions when there are unknown health risks for the woman and the fetus during pregnancy, epigenetic effects, and health issues that may not manifest until adulthood or old age (or even in future generations)? It would be extremely difficult, if not impossible, to conduct the type of follow-up studies required to conclude that human genome editing is safe enough for use in reproduction.
Human germline editing is more than a scientific or technological issue. It has an impact on how we understand ourselves as humans and the kind of future we want to create. It has far-reaching implications for society as a whole, not just for individuals. As a result, decisions about whether or not to allow germline modification should not be made by small groups of scientists or bioethicists, biotechnology companies, or wealthy elites. Human germline editing is an urgent social justice issue; we need open public discussions about it.