Simple hydrocarbons for use as refrigerants are comfortably inside it. There has been a move towards hydrocarbons and away from chlorofluorocarbons (CFCs) as refrigerants because of harm done by the latter to the ozone layer
Additionally to ethane the following simple hydrocarbon compounds find use as refrigerants [4]: propane (R290), n-butane (R600), isobutane (R600a) and propylene (R1270). The last three of these have normal boiling points respectively 273, 261 and 225 K. The refrigerating effect is expressed as: Tc (Sg – Sf)
where Tc (K) is the cold enclosure temperature and Sg and Sf (J Kg-1 K-1) are the specific entropies of respectively refrigerant vapour at liquid at the higher surrounding temperature Th. We expect intuitively, working on a trouton’s constant’ basis, that this could be re-written:
Refrigerating effect = Δhvap Tc/Th J kg-1
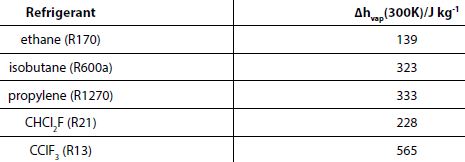
where Δhvap (J kg-1) is the heat of vaporisation at Th. Imagine then a refrigerating device operating under conditions such that Th = 300K. The values of Δh at this temperature for a selection of refrigerants — rap hydrocarbons and CFC – are given in the table below.