Researchers are developing tougher circuits to help them withstand the rigors of energy production, space exploration, and other endeavors. Electronics that can withstand extreme temperatures will be required for tomorrow’s cutting-edge technology. That’s why, right now, a team of researchers led by Michigan State University’s Jason Nicholas is constructing stronger circuits.
With the help of nickel, Nicholas and his team have created more heat-resistant silver circuitry. The research, which was funded by the US Department of Energy Solid Oxide Fuel Cell Program, was published on April 15 in the journal Scripta Materialia.
The MSU team’s efforts to benefit next-generation fuel cells, high-temperature semiconductors, and solid oxide electrolysis cells could have applications in the automotive, energy, and aerospace industries.
Although these devices are not yet available for purchase, researchers are currently developing them in laboratories for testing in the real world and even on other planets. On April 22, for example, NASA developed a solid oxide electrolysis cell that allowed the Mars 2020 Perseverance Rover to produce oxygen from gas in the Martian atmosphere. NASA hopes that this prototype will one day lead to equipment that will allow astronauts on Mars to create rocket fuel and breathable air.
Researchers are building tougher circuits to help withstand the grueling demands of energy production, space exploration and more.
However, in order for such prototypes to become commercial products, they must be able to perform at high temperatures for extended periods of time, according to Nicholas, an associate professor in the College of Engineering. He was drawn to this field after years of using solid oxide fuel cells, which function in reverse to solid oxide electrolysis cells. Instead of using energy to produce gases or fuel, they generate energy from those chemicals.
“Solid oxide fuel cells operate on high-temperature gases. We can electrochemically react those gases to produce electricity, which is far more efficient than exploding fuel as an internal combustion engine does “Nicholas, who directs a lab in the Department of Chemical Engineering and Materials Science, explained.
Even in the absence of explosions, the fuel cell must be able to withstand harsh working conditions. “These devices commonly operate at temperatures ranging from 700 to 800 degrees Celsius, and they must do so for an extended period of time — 40,000 hours over their lifetime,” Nicholas explained. That’s roughly 1,300 to 1,400 degrees Fahrenheit, or roughly double the temperature of a commercial pizza oven.
“And over that lifetime, you’re thermally cycling it,” Nicholas said. “You’re cooling it down and heating it back up. It’s a very extreme environment. You can have circuit leads pop off.” Thus, one of the hurdles facing this advanced technology is rather rudimentary: The conductive circuitry, often made from silver, needs to stick better to the underlying ceramic components.
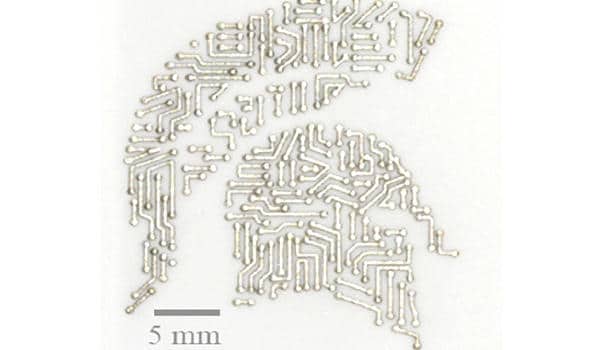
The researchers discovered that adding an intermediate layer of porous nickel between the silver and the ceramic improved adhesion. The team optimized how the nickel was deposited on the ceramic by conducting experiments and computer simulations of how the materials interact. The researchers used screen printing to create the thin, porous nickel layers on the ceramic in a pattern or design of their choice.
“It’s the same screen printing used to make T-shirts,” Nicholas explained. “Instead of shirts, we’re screen-printing electronics. It’s a very efficient manufacturing method.”
Once the nickel is in place, the team exposes it to silver that has been melted at a temperature of around 1,000 degrees Celsius. The nickel not only withstands the heat (its melting point is 1,455 degrees Celsius), but it also uses capillary action to distribute the liquified silver uniformly over its fine features.
“It almost looks like a tree,” Nicholas remarked. “Capillary action allows water to reach the branches of a tree. Through the same mechanism, the nickel is wicking up the molten silver.”
Even in the 700 to 800 degree Celsius heat of a solid oxide fuel cell or a solid oxide electrolysis cell, the nickel keeps the silver locked onto the ceramic as it cools and solidifies. In addition, this approach has the potential to benefit other technologies where electronics can overheat.
“A wide range of electronic applications necessitate circuit boards that can withstand high temperatures or high power,” said Jon Debling, a technology manager with MSU Technologies, Michigan State’s tech transfer and commercialization office. “These include both existing applications in the automotive, aerospace, industrial, and military markets, as well as newer ones like solar cells and solid oxide fuel cells.”
As a technology manager, Debling works to commercialize Spartan innovations, and he is assisting in the patenting of this process for making tougher electronics. “This technology is a significant improvement over existing paste and vapor deposition technologies in terms of cost and temperature stability,” he said.
Nicholas, for one, remains most interested in the cutting-edge applications that are on the horizon, such as solid oxide fuel cells and solid oxide electrolysis cells. “We’re working to improve their dependability both here on Earth and on Mars,” Nicholas explained.