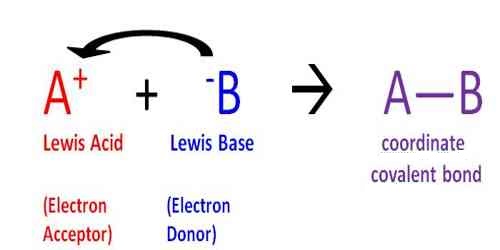
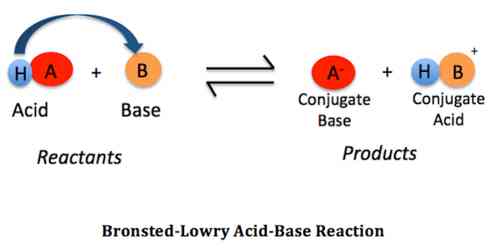
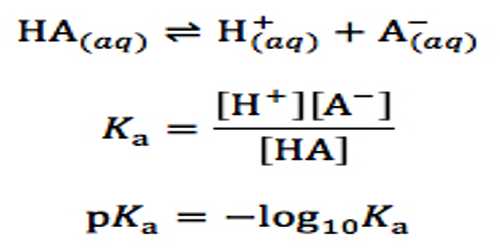Chemistry
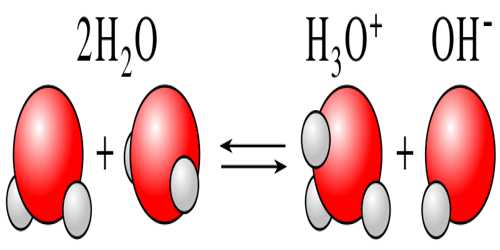
Dissociation of weak acids and weak bases: Ostwald’s Dilution Law
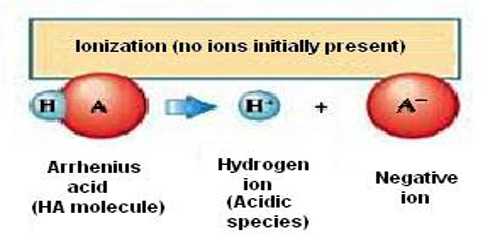
Dissociation of weak acids and weak bases: Ostwald’s Dilution Law Weak electrolytes are incompletely dissociated into ions. Ostwald’s dilution law is the application of the…