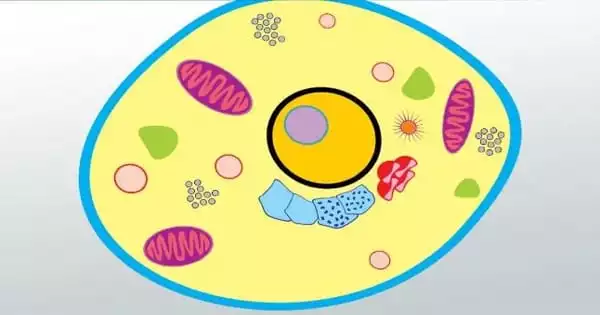
Embryonic stem cells and other pluripotent cells divide fast and can differentiate into practically any cell type in the body. Scientists have long attempted to decipher the signals that cause stem cells to lose their pluripotency and adopt their final functional state. Researchers say in a new study that they have found a critical regulator of this process, a molecule known as BEND3.
Researchers describe identifying a crucial regulator of this process in a paper published in the Proceedings of the National Academy of Sciences. They discovered that a protein known as BEND3 inhibits the expression of hundreds of genes involved in differentiation, allowing the cell to retain its stem cell-like characteristics. Only after BEND3 is suppressed do cells take on their final form and function. They normally stop actively reproducing after they have differentiated.
The discoveries are important for understanding normal development and may be valuable in cancer research, according to Supriya Prasanth, professor and department chair of cell and developmental biology at the University of Illinois Urbana-Champaign.
Stem cell therapy may be able to regenerate damaged tissue. Stem cells must develop into the target cell, reach the site of injury, survive, and engraft in order to reach their full therapeutic potential. Cell morphology, lineage specific markers, cell specific function, and gene expression must all be evaluated to adequately characterize these cells. Cell fate imaging is critical for monitoring survival and engraftment. Only then can the effectiveness of therapy be determined by evaluating organ specific function.
Most tumors have this rapid multiplication because cell-cycle regulators aren’t working properly. The prognosis of how cancer cells will respond to treatment is frequently related to their differentiation status. The better the prognosis, the more differentiated the tumor.
Professor Supriya Prasanth
“Most tumors have this rapid multiplication because cell-cycle regulators aren’t working properly,” she explained. “The prognosis of how cancer cells will respond to treatment is frequently related to their differentiation status. The better the prognosis, the more differentiated the tumor.”
According to Prasanth, stem cells have the ability to repopulate a cancer tumor that has shrunk during treatment. Finding a genetic switch that shifts cancer cells from proliferation to differentiation could help with cancer treatment.
Prasanth’s lab studies cell cycle regulators. Her preliminary research revealed BEND3 as a potentially essential system player. Her team discovered that binding BEND3 to certain locations along the chromosome lowered or prevented the expression of dozens of genes. When BEND3 was eliminated, gene expression returned to normal.
“When you do gene-expression studies, you might see hundreds of genes go up and hundreds of genes go down,” Prasanth explained. “But what exactly does it mean?”
She and her colleagues discovered that several of the genes suppressed by BEND3 boost cell differentiation in their recent study. Graduate students Fredy Kurniawan and Neha Chetlangia led the research with postdoctoral researcher Mohammad Kamran, in collaboration with the laboratories of University of Illinois cell and developmental biology professor Kannanganattu Prasanth and Mirit Aladjem, a senior investigator at the National Cancer Institute.
“BEND3 binding to these genes inhibits their expression, preventing cells from acquiring a differentiated state,” Supriya Prasanth explained. “And the instant that control is removed, the cells begin to proceed toward the differentiation route.”
According to Supriya Prasanth, BEND3 is not the only regulator of the cell-differentiation pathway; it binds to and interacts with numerous other molecular regulators of this process. However, its presence or absence appears to be important in determining a cell’s fate, making it an appealing target for prospective medicinal interventions if the process goes wrong.
The Supriya Prasanth lab and collaborators at the Memorial Sloan Kettering Cancer Center revealed structural insights into BEND3-mediated gene regulation in an accompanying publication published in the journal Genes and Development.
Cells must differentiate in order to specialize for diverse functions in order to form multicellular creatures. The mammalian body is made up of three types of cells: germ cells, somatic cells, and stem cells. Except for certain cell types, such as red blood cells, which lack nuclei in their fully developed stage, each of the approximately 100 trillion cells in an adult person possesses its own copy or copies of the genome. The majority of cells are diploid, meaning they have two copies of each chromosome.
The process of cellular differentiation is governed by transcription factors and growth factors, and it results in the expression or repression of distinct genes amongst cell types, resulting in different proteomes. The difference in proteomes between cell types is what causes differentiation and ultimately cell specialization. The ability of transcription factors to govern whether or not a gene is transcribed, which leads to specialization, and the ability of growth factors to aid in the division process are important components of cell differentiation.