Intensive and extensive thermodynamic properties
Thermodynamic properties of a system may be classified into two groups:
- Intensive properties and
- Extensive properties
Any property of a system that does not depend on the actual quantity of matter contained in the system is called Intensive property. Pressure, temperature, boiling point, melting point, density, concentration etc., are examples of Intensive properties. A particularly important type of intensive property is the specific property, which is always given on a unit mass basis. An example is specific volume, which has units of volume/mass, typically expressed as cubic feet per pound or cubic meters per kilogram.
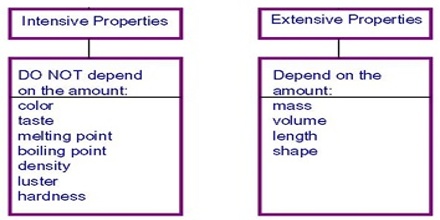
On the other hand, the property of a system that is dependent upon the actual quantity of matter contained in the system is called extensive property. Mass, volume, enthalpy, internal energy, entropy, number of moles etc are examples of extensive properties. Volume is an example. If you double the length of all edges of a solid cube, the volume increases by a factor of eight.
Extensive properties are additive, while the intensive properties are non-additive.