Nanoplasmonic imaging is a powerful technique that enables researchers to observe real-time biological events at the nanoscale. It involves the use of metallic nanoparticles, which can interact with light in a way that is highly sensitive to the local environment. When a protein molecule binds to a nanoparticle, it causes a detectable change in the way the light is scattered or absorbed by the nanoparticle.
A nanoplasmonics approach was used by researchers to observe the real-time production of cell secretions, including proteins and antibodies; this advancement could aid in the development of cancer treatments, vaccines, and other therapies.
Cell secretions such as proteins, antibodies, and neurotransmitters are critical in immune response, metabolism, and cell communication. Understanding cell secretions is critical for developing disease treatments, but current methods can only report the number of secretions produced, with no information on when and where they are produced.
Now, researchers in the BIOnanophotonic Systems Laboratory (BIOS) in the School of Engineering and at the University of Geneva have developed a novel optical imaging approach that gives a four-dimensional view of cell secretions in both space and time. By placing individual cells into microscopic wells in a nanostructured gold-plated chip, and then inducing a phenomenon called plasmonic resonance on the chip’s surface, they are able to map secretions as they are being produced while observing cell shape and movement.
Cell secretions are like the words of the cell: they spread out dynamically in time and space to connect with other cells. Our technology captures key heterogeneity in terms of where and how far these ‘words’ travel.
Saeid Ansaryan
The scientists believe their method, which was recently published in Nature Biomedical Engineering, has “tremendous” potential for pharmaceutical development as well as fundamental research because it provides an unprecedentedly detailed view of how cells function and communicate.
“One of the most important aspects of our work is that it allows us to screen cells individually in a high-throughput manner. Collective measurements of many cells’ average responses do not reflect their heterogeneity… Everything in biology is heterogeneous, from immune responses to cancer cells. This is why cancer is so difficult to treat “Hatice Altug, CEO of BIOS.
A million sensing elements
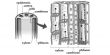
At the heart of the scientists’ method is a 1 cm2 nanoplasmonic chip composed of millions of tiny holes, and hundreds of chambers for individual cells. The chip is made of a nanostructured gold substrate covered with a thin polymer mesh. Each chamber is filled with a cell medium to keep the cells alive and healthy during imaging.

“Cell secretions are like the words of the cell: they spread out dynamically in time and space to connect with other cells. Our technology captures key heterogeneity in terms of where and how far these ‘words’ travel,” says BIOS PhD student and first author Saeid Ansaryan.
The nanoplasmonics part comes in thanks to a light beam, which causes the gold electrons to oscillate. The nanostructure is engineered so that only certain wavelengths can penetrate it. When something — like protein secretion — occurs on the chip’s surface to alter the light passing through, the spectrum shifts. A CMOS (Complementary Metal Oxide Semiconductor) image sensor and an LED translate this shift into intensity variations on the CMOS pixels.
“The genius of our device is that the nanoholes distributed across the entire surface turn every spot into a sensing element. This enables us to observe spatial patterns of released proteins regardless of cell position “Ansaryan explains.
The method enabled the scientists to observe two critical cellular processes, cell division and cell death, as well as study delicate antibody-secreting human donor B-cells.
“The cell content released during two types of cell death, apoptosis and necroptosis, was observed. The content is released in an asymmetric burst in the latter, resulting in an image signature or fingerprint. This has never been demonstrated before at the single-cell level “Altug states.
Screening for cell fitness
The cells under study can be easily recovered because the method bathes them in a nutritious cell medium and does not require the toxic fluorescent labels used by other imaging technologies. This gives the method a lot of potential for use in the development of pharmaceutical drugs, vaccines, and other treatments, such as helping researchers understand how cells respond to different therapies at the individual level.
“Because the amount and pattern of secretions produced by a cell are a proxy for determining their overall effectiveness,” says Ansaryan, “we could also imagine immunotherapy applications where you screen patient immune cells to identify the most effective, and then create a colony of those cells.”