We find many non-engineered metals to be incredibly tough and durable. And while it is true that these forged materials are technically very rigid – they do not break or fracture when applied to the force – and very strong – a high level of force must be applied to them before they can be distorted – both pure metal and even our best Alloys, like steel, lack strength. To the physicist, hardness refers to the ability of a substance to stay together when applying friction.
Metals do not hold well in this case; In engines and other mechanical devices where metal parts are crushed against other metal parts, the friction causes the outer layers to move away over time. This is why oily protective lubricants are used in gears and pistons and why saws and screws have diamond-tipped blades and bits.
Engineers in electronics with metal-to-metal connections often cover inexpensive metals with more natural resistant precious metal alloys to increase the longevity of the device; Even then, this cover will eventually be scrapped due to the speed of repetition.
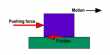
So far, a team of researchers at Sandia National Laboratories has developed a new type of metal that breaks the world record for wear resistance. The results of their analysis published in Advanced Materials show that their 9 to 1 platinum-gold alloy is 100 times more durable than high-steel. It is the first all-metallic material to show comparable wear rates with carbon (DLC), such as strong known materials, sapphires, and diamonds.
In laboratory experiments where the friction track of the metal stainless steel was repeatedly extended, the mixed surface showed negligible signs of structural change after scanning electron microscope imaging after 100,000 sliding passes.
Chris Nordquist, a Sandia engineer not involved in the study, said in a statement, “These wear-resistant materials can provide reliability benefits for many of the devices we’ve explored.” “Opportunities for integration and improvement will be device-specific, but this component will provide another tool to address the current reliability limitations of metallic micro-electronic components.”
The Sandia team notes that the impressive properties of 9 to 1 platinum-gold alloys are already well documented. The creation of exceptional resistance to their formation comes from the unique method they create: a very thin layer of platinum -gold is deposited on top of a high-purity platinum-gold base using a vacuum-sealed magnetic chamber at once.
The inspiration for using this fictional method came from computer models of how individual atoms would interact. Although stability under mechanical stress was predicted by their simulations, the platinum-gold alloy also had some unexpected advantages. In an experiment recorded in the Carbon Journal, during the wear test the components spontaneously form a coating of their own lubricant – DLC. DLC is one of the best lubricants because it has the hardness of diamond but also the slickness of graphite.
First author John Curry explained, “We believe that carbon dioxide molecules from the environment can eventually trap and reduce carbon dioxide molecules from sliding into a diamond-like carbon formation as a result of wear resistance and innate resistance.” “There are other ways to do this in the industry but they are usually involved in vacuum chambers with high-temperature plasmas of carbon species. It can be very expensive. “