Ethylene (CH2 =CH2) is obtained by cracking petroleum distillate and is an important ‘building block’ in petrochemical manufacture. Large quantities of it are transported. Ethylene has a critical temperature of 9°C.
At that temperature its vapour pressure is approximately 50 bar. The vapour pressure can be brought down by refrigeration and this is the way in which ethylene is usually transported by sea. The vapour pressure will be above atmospheric as the temperature will be above the normal boiling point, therefore ethylene in such a state is a refrigerated liquefied gas, an expression not encountered previously in this volume. This is a preferable term to ‘cryogenic ethylene’ which is often encountered and which would only be accurate if the ethylene was cooled all the way down to its normal boiling point of -104°C.
We saw previously that LPG has a vapour pressure at room temperature of about 8 bar. Ethylene has no `vapour pressure at room temperature’ because of its low critical temperature. If, as sometimes happens, a stationary or marine tank for LPG is to be used for ethylene the vapour pressure of the ethylene can by refrigeration be brought down to that for which the vessel was designed. Refrigeration down to -50 to -60°C will be required bring the vapour pressure down to that of an LPG payload without refrigeration.
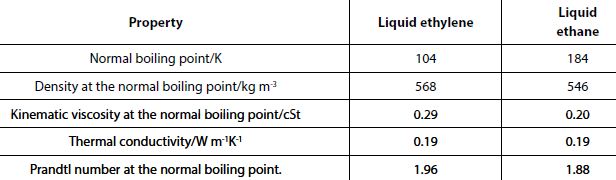
The normal boiling point of ethylene (104K) is close to that of methane (112K). That of ethane (184K) falls between that methane and that of propane (231K). Some of the properties for methane and propane given in the previous table are given for ethylene and ethane in the table below.