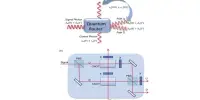
Quantum computers have demonstrated promise in replicating and comprehending the atomic dynamics of light-sensitive molecules such as those engaged in photosynthesis or photovoltaics. These molecules are essential in energy conversion processes and a variety of chemical reactions.
Duke University researchers used a quantum-based approach to detect a quantum effect in the way light-absorbing molecules interact with incoming photons. The result, known as a conical intersection, limits the paths molecules can take to switch between distinct forms.
The observation approach employs a quantum simulator generated through quantum computing research, and it solves a long-standing, fundamental challenge in chemistry that is crucial to processes like photosynthesis, vision, and photocatalysis. It is also an illustration of how developments in quantum computing are being applied to fundamental science research.
The findings are published in the journal Nature Chemistry.
“As soon as quantum chemists ran into these conical intersection phenomena, the mathematical theory said that there were certain molecular arrangements that could not be reached from one to the other,” said Kenneth Brown, the Michael J. Fitzpatrick Distinguished Professor of Engineering at Duke. “That constraint, known as a geometric phase, is not impossible to measure, but no one has been able to do so. Using a quantum simulator allowed us to see it in its natural quantum state.”
As soon as quantum chemists ran into these conical intersection phenomena, the mathematical theory said that there were certain molecular arrangements that could not be reached from one to the other.
Kenneth Brown
Conical intersections govern electron migration between energy levels and can be represented as a mountain top contacting the point of its reflection from above. The energy levels and physical locations of an unexcited molecule in its ground state are represented by the bottom half of the conical intersection. The top half is the identical molecule, but with its electrons activated as a result of absorbing energy from an incoming light particle.
The molecule can’t stay in the top state because its electrons are out of alignment with their host atoms. To return to the lower energy state, the atoms of the molecule begin rearranging themselves to meet the electrons. The conical intersection where the two mountains meet signifies an inflection point. The atoms can either fail to get to the other side by readjusting to their original state, dumping excess energy in the molecules around them in the process, or they can successfully make the switch.
However, because atoms and electrons move so quickly, they exhibit quantum effects. The molecule is in numerous shapes at once, rather than being in any one shape – at any one location on the mountain – at any given time. All of these potential sites may be symbolized by a blanket draped across a piece of the mountainous landscape.
Certain molecular transformations, however, are not possible due to a mathematical quirk in the system that derives from the underlying mathematics, known as a geometric phase. The blanket cannot completely encircle the peak.
“If a molecule has two different paths to take to get to the same final shape, and those paths happen to surround a conical intersection, then the molecule wouldn’t be able to take that shape,” Jacob Whitlow, a PhD student working in Brown’s research, explained. “It’s an effect that’s hard to gain intuition for, because geometric phase is weird even from a quantum mechanical standpoint.”
Measuring this quantum effect has always been difficult because it is both short-lived (femtoseconds) and small (atom-scale). And any disruption to the system will prevent it from being measured. While academics have examined and analyzed numerous smaller components of the greater conical junction phenomena, the geometric phase has always eluded them.
“If conical intersections exist – which they do – then the geometric phase has to exist,” said Brown, who also holds appointments in Duke physics and chemistry. “But what does it mean to say something exists that you can’t measure?”
In the paper, Whitlow and coworkers used a five-ion quantum computer built by the group of Jungsang Kim, the Schiciano Family Distinguished Professor of Electrical and Computer Engineering at Duke. The quantum computer uses lasers to manipulate charged atoms in a vacuum, providing a high level of control. Whitlow and Zhubing Jia, a PhD student in Brown’s laboratory, also expanded the capability of the system by developing ways to physically nudge the floating ions within their electromagnetic traps.
Based on how the ions are moved and the quantum state in which they are placed, they can fundamentally display the same quantum mechanisms as the motion of atoms around a conical intersection. Because the quantum dynamics of the trapped ions are nearly a billion times slower than those of a molecule, the researchers were able to take direct measurements of the geometric phase in action.
The results resemble a two-dimensional crescent moon. As shown in the conical intersection graph, certain configurations on one side of the cone fail to reach the other side of the cone despite the absence of an energy barrier. The experiment, Brown says, is an elegant example of how even today’s rudimentary quantum computers can model and reveal the inner quantum workings of complex quantum systems.
“The beauty of trapped ions is that they get rid of the complicated environment and make the system clean enough to make these measurements,” Brown went on to say.
An ion trap quantum simulator was used in an independent experiment at the University of Sydney in Australia to observe the effects of the geometric phase. Many technical specifics change between approaches, but the main observations are consistent. The Sydney study will appear in the same edition of Nature Chemistry.