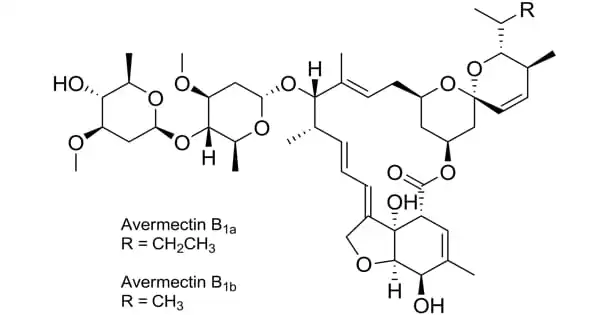
Abamectin is an insecticide and anthelmintic that is widely used. It is a 16-membered ring macrolide molecule discovered by Kitasato University in Japan and Merck Company (United States). It is an Avermectin family member and a natural fermentation product of the soil-dwelling actinomycete Streptomyces avermitilis. It’s insecticidal, acaricidal, and nematicidal.
Abamectin (also known as Avermectin B1) varies from Ivermectin, the most well-known member of the Avermectin family, by having a double bond on C-22-25. Streptomyces avermitilis fermentation produces eight closely similar Avermectin homologues, with B1 (a and b) accounting for the majority of the fermentation. Abamectin is a nonproprietary term for a combination of Bla (80%) and B1b (20%). Abamectin is the only Avermectin that is utilized in both agriculture and pharmaceuticals.
Properties
- Melting point: 150-155°C
- Boiling point: 717.52°C (rough estimate)
- Density: 1.16
- Flash point: 150 °C
- Storage temp.: Sealed in dry,Store in freezer, under -20°C
- Solubility: Soluble in DMSO
- Water Solubility: 0.007-0.01 mg l-1 (20 °C)
- Form: neat
The fermentation of Streptomyces avermitilis produces it. Natural abamectin contains eight components, four of which are major components, namely A1a, A2a, B1a, and B2a, with a total content of ≤80%; the remaining four components, corresponding to a smaller percentage, are A1b, A2b, B1b, and B2b, with a total content of ≤20%. The major insecticidal ingredient in currently sold Abamectin insecticide is abamectin (Abamectin B1a + B1b, with B1a being at least 90% and B1b being less than 5%).
Mode of Action
Avermectins bind to glutamate-gated chloride channels present in nerve and muscle cells of invertebrates. They induce these cells to hyperpolarize, resulting in paralysis and death. Mammalian glutamate-gated chloride channels are found only in the brain and spinal cord, and because Avermectins have a low affinity for other mammalian ligand-gated channels and do not typically cross the blood–brain barrier, they are very safe for mammals.
History
Following an intense search for natural compounds having anthelmintic action, avermectins were identified in 1967 in fermentation broths of an actinomycete culture obtained from the Kitasato Institute in Japan. The United Nations did not employ ivermectin to treat Onchocerca volvulus (Onchocerciasis or River blindness) in people until 1985. Avermectin’s discoverers, William C. Campbell and Satoshi Mura, shared the 2015 Nobel Prize in Physiology or Medicine.
Activity
Abamectin is an insecticide as well as an acaricide (miticide) and a nematicide. It is also used to control fire ants. Abamectin is provided orally to horses for deworming them.
Use
Abamectin is also used in veterinary medicine as an antihelmintic. Although resistance to abamectin-based antihelmintics is increasing, it is not as common as resistance to other types of veterinary antihelmintics. Emamectin benzoate, a benzoate salt, is also employed as an insecticide. Avermectins have been used to treat parasitic diseases in both humans and animals. Avermectins, including abamectin, have been investigated as anti-alcohol therapy. Ivermectin has recently been explored for its potential use as an anti-inflammatory agent.