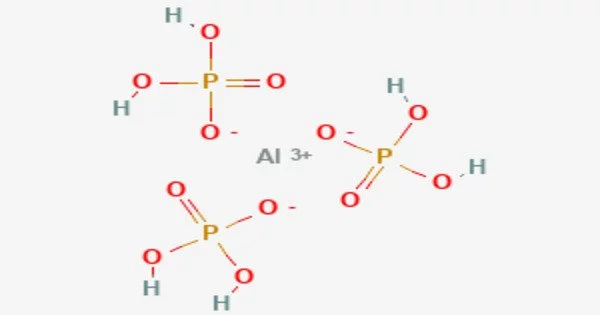
Aluminum dihydrogen phosphate is an inorganic compound with the chemical formula Al(H2PO4)3. It is a white crystalline powder that is used as a food additive as a leavening agent or a gelling agent. It is also used in the pharmaceutical industry as a filler or excipient in tablets and capsules.
Aluminium dihydrogenphosphate describes inorganic compounds with the formula Al(H2PO4)3.xH2O where x = 0 or 3. They are white solids. Upon heating these materials convert sequentially to a family of related polyphosphate salts including aluminum triphosphate (AlH2P3O10.2H2O), aluminium hexametaphosphate (Al2P6O18), and aluminium tetrametaphosphate (Al4(P4O12)3). Some of these materials are used for fireproofing and as ingredients in specialized glasses.
Properties
It is a chemical compound. It is also known as aluminum monohydrogen phosphate or aluminum phosphate. It is a white crystalline solid that is soluble in water. Some physical properties of aluminum dihydrogen phosphate include a molecular weight of 243.98 g/mol and a density of 2.5 g/cm3. This compound is commonly used in the food industry as an emulsifying agent and a leavening agent.
- Chemical formula: AlH6O12P3
- Molar mass: 317.939 g·mol−1
- Appearance: white solid
- Density: 2.37 g/cm3

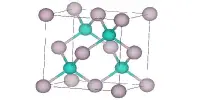
According to analysis by X-ray crystallography, the structure consists of a coordination polymer featuring octahedral Al3+ centers bridged by tetrahedral dihydrogen phosphate ligands. The dihydrogen phosphate ligands are bound to Al3+ as monodentate ligands.
Applications
The compound is used in various industrial and medicinal applications, such as in the production of catalysts, fireproofing materials, and as a phosphorus source in fertilizers. It is an inorganic salt that is used in various applications, including as a food additive, a flame retardant, and in the production of ceramics and glass.