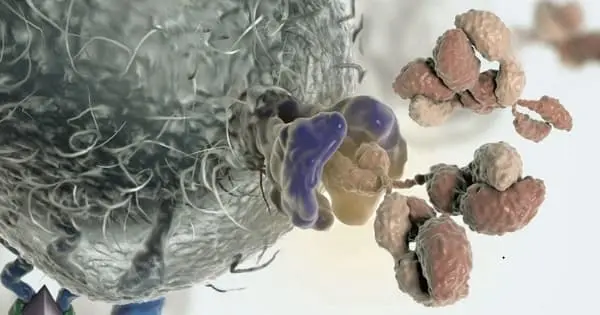
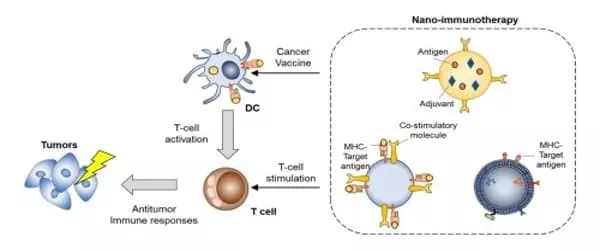
Cancer immunotherapy is a cancer treatment strategy that trains the body’s immune system to recognize, control, and eliminate tumor cells. The use of nanoparticles (NPs) for precise and timely delivery of immunotherapies to specifically targeted neoplasms, with little or no harm to healthy cells, maximizes immunotherapy efficacy.
In clinical settings, cancer immunotherapy is an appealing treatment option. The major challenges of immunotherapy, however, are limited patient response, tumor specificity, immune-related adverse events, an immunosuppressive tumor microenvironment. As a result, nanoparticle (NP)-based drug delivery has been used to not only improve the efficacy of immunotherapeutic agents but also to significantly reduce toxicity.
Wake Forest School of Medicine researchers discovered that a nanoparticle therapeutic improves cancer immunotherapy and could be a new approach to treating malignant pleural effusion (MPE). MPE is characterized by the accumulation of fluid between the chest wall and the lungs, as well as the presence of malignant cells and/or tumors.
The study’s findings are published in the current issue of Nature Nanotechnology. Each year, more than 200,000 new cases of MPE are diagnosed in the United States, with non-small cell lung cancer accounting for more than one-third of all cases.
Clinical evidence also suggests that MPE contains an abundance of tumor-associated immune cells, which prevent the body’s immune system from recognizing and eliminating cancer. This ‘cold’ immune environment may have played a significant role in ICI’s failure.
Dawen Zhao
“MPE is associated with a poor prognosis, with an average survival of only four to nine months,” said Dawen Zhao, M.D., Ph.D., associate professor of biomedical engineering at Wake Forest School of Medicine. “MPE can also have a negative impact on quality of life because it causes shortness of breath, pain, weight loss, and decreased physical activity.”
According to Zhao, recent clinical trials involving immune checkpoint inhibitors (ICI) or novel immunotherapies such as anti-PD-1 have yielded promising results in MPE patients. However, immunotherapy benefits only a small number of MPE patients, and many suffer from immunotoxicity.
“Clinical evidence also suggests that MPE contains an abundance of tumor-associated immune cells, which prevent the body’s immune system from recognizing and eliminating cancer,” Zhao explained. “This ‘cold’ immune environment may have played a significant role in ICI’s failure.”
Zhao and his colleagues created a nanoparticle called liposomal cyclic dinucleotide (LNP-CDN) for targeted activation of an immune pathway called STING, which reprograms tumor-associated immune cells to active anti-tumor ones.
MPE is frequently associated with two distinct compartments within the tumor microenvironment, the effusion and pleural tumors, both of which coexist within the pleural cavity. Therapeutic interventions and drug delivery are complicated by these two distinct compartments.
In a mouse model, intrapleural injection reduced the ‘cold’ immune environment not only in the effusion space, but also within the tumors. Zhao’s team combined LNP-CDN with anti-PD-L1 immunotherapy, which significantly reduced the volume of MPE and inhibited tumor growth in both the pleural cavity and lung tissue in MPE mice, resulting in prolonged survival.
Zhao’s team also tested the nanoparticle therapeutic on human MPE tissue samples, and the results were similar: increased tumor cell killing by cytotoxic immune cells. “Whether administered alone or in combination with immunotherapy, this study demonstrates a potential treatment for MPE,” Zhao said. “Given the current prognosis of MPE patients, new interventions are required not only to extend survival but also to improve quality of life.”
Immunotherapy has been clinically validated as the first line of treatment and management for the majority of malignancies, and it may also be an effective therapy for patients with specific tumors that are resistant to treatment. Regardless of the fact that immunotherapy can be administered to patients in a variety of ways, transporting small-particle medications to their malignant growth target destinations of activity generally presents the most difficulty due to the cancer’s homogeneous tissue dispersion, renal freedom, and lack of target specificity.
Recently, advanced NP-based drug delivery systems were successfully used in cancer immunotherapy to reduce toxic side effects and immune-related adverse events. Repurposing these NPs as immunotherapeutic agent delivery systems has the potential to overcome the limitations of current cancer immunotherapy. Here, we focus on recent advances in NP-based immunotherapeutic delivery systems, such as ICD-inducing drugs, cytokines, and adjuvants for promising cancer immunotherapy.