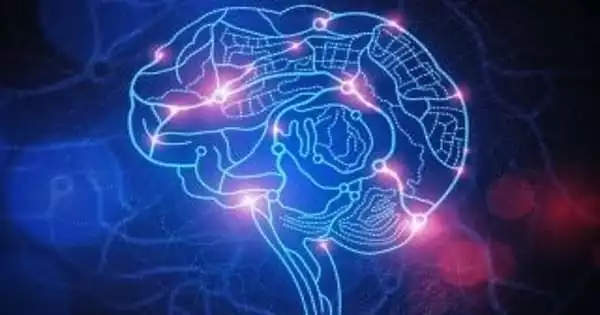
According to a study published today in eLife, normal brain aging is accelerated by around 26 percent in adults with progressing type 2 diabetes compared to individuals without the disease. The authors looked at the association between normal brain aging and type 2 diabetes and discovered that type 2 diabetes has a similar pattern of neurodegeneration as aging but proceeds faster. One key implication of this discovery is that even normal brain aging may reflect changes in the brain’s glucose control by insulin.
The findings also show that by the time type 2 diabetes is properly diagnosed, severe anatomical damage to the brain has already occurred. Sensitive methods of detecting diabetes-related brain alterations are thus urgently required.
Despite the fact that there is considerable evidence relating type 2 diabetes with cognitive deterioration, few patients now receive a full cognitive assessment as part of their therapeutic care. It might be difficult to tell the difference between natural brain aging, which starts in middle life, and brain aging caused or hastened by diabetes. No studies have directly compared brain changes in healthy people over the course of their lives to changes in people of the same age with diabetes.
Our findings suggest that type 2 diabetes and its progression may be associated with accelerated brain aging, possibly due to compromised energy availability causing significant changes in brain structure and function.
Lilianne Mujica-Parodi
“Routine clinical assessments for diagnosing diabetes typically focus on blood glucose, insulin levels and body mass percentage,” says first author Botond Antal, a PhD student at the Department of Biomedical Engineering, Stony Brook University, New York, US. “However, the neurological effects of type 2 diabetes may reveal themselves many years before they can be detected by standard measures, so by the time type 2 diabetes is diagnosed by conventional tests, patients may have already sustained irreversible brain damage.”
The team used the largest available brain structure and function dataset spanning the human lifespan to identify the impact of diabetes on the brain above and beyond normal aging: UK Biobank data from 20,000 adults aged 50 to 80 years old. This dataset contains brain scans and brain function assessments for both healthy and type 2 diabetic subjects. They utilized this to establish which brain and cognitive changes are particular to diabetes rather than simply aging, and then validated their findings by comparing them to a meta-analysis of over 100 additional studies.
Both aging and type 2 diabetes, according to their findings, produce alterations in executive functions such as working memory, learning, and flexible thinking, as well as changes in brain processing speed. People with diabetes, on the other hand, experienced an additional 13.1 percent decline in executive function beyond age-related effects, and their processing speed reduced by 6.7 percent compared to people of the same age who did not have diabetes. Their meta-analysis of other studies verified this finding: people with type 2 diabetes had consistently and significantly worse cognitive function than healthy individuals of the same age and education.
MRI images were also used to assess brain anatomy and activity in patients with and without diabetes. They discovered a loss in grey brain matter with age, mainly in an area known as the ventral striatum, which is important for the brain’s executive processes. People with diabetes, on the other hand, showed even more dramatic losses in gray matter beyond the standard age-related effects — a further 6.2 percent decline in grey matter in the ventral striatum, but also loss of grey matter in other locations, when compared to normal aging.
The findings imply that the patterns of neurodegeneration associated with type 2 diabetes significantly overlap with those of normal aging, but that neurodegeneration is accelerated. Furthermore, the impairments on brain function became more severe as diabetes duration rose. Diabetes progression has been associated to a 26% acceleration of brain aging.
“Our findings suggest that type 2 diabetes and its progression may be associated with accelerated brain aging, possibly due to compromised energy availability causing significant changes in brain structure and function,” says senior author Lilianne Mujica-Parodi, Director of Stony Brook University’s Laboratory for Computational Neurodiagnostics. “This harm may have already happened by the time diabetes is properly diagnosed. However, brain imaging could give a clinically useful parameter for diagnosing and monitoring the neurocognitive effects of diabetes. Our findings highlight the importance of research into brain-based biomarkers for type 2 diabetes, as well as therapy options that explicitly target its neurocognitive impacts.”