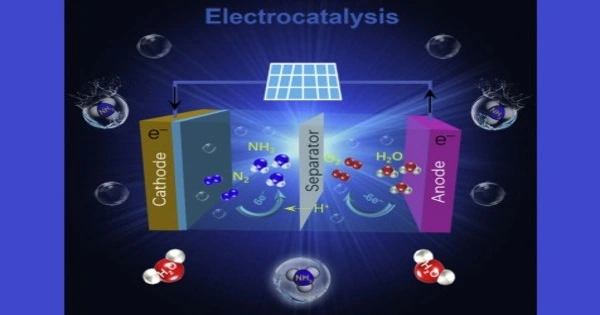
Electrochemistry has proven to be a potent tool for organic synthesis, and a rising number of prominent synthetic chemists are investigating the combining of standard synthetic techniques with electrochemistry, a process known as “electrifying synthesis.” Chemical reductants and oxidants are not required, but electrons from/to an electrical power source can be used for organic reduction and oxidation (redox) reactions, which helps to sustainable development goals in part (SDGs).
Electrifying synthesis has become a popular catchphrase among synthetic chemists. In addition to the traditional two- or three-electrode systems that use batch-type cells, recent advances in organic electrochemical processes, such as microflow electrochemical reactors, Li-ion battery-like technologies, and bipolar electrochemistry, have been noteworthy.
If electrochemical reactions for organic compound synthesis were power supply independent, they would have a broader range of uses. Researchers have now used the flow of electrolytes in an electrochemical reactor to provide the streaming potential for reactions that do not require electricity. They successfully polymerized two aromatic monomers, demonstrating the setup’s viability.
Many manufacturing businesses rely on the synthesis of organic chemicals and polymers. The new “electrifying synthesis” technologies, which combine traditional synthetic chemistry with electrochemistry, bring us one step closer to a more sustainable future. These reactions do not necessitate the use of potentially hazardous chemical reagents. They produce organic synthesis by simply conducting redox reactions using electrons from an electric power source.
We have seen a huge leap in the development of electrochemical reactors for carrying out organic synthesis, but most of them require a power source. We wanted to build a power-independent system to make the process more accessible. And we found the answer to our quest in streaming potential-driven electrochemistry.
Prof. Shinsuke Inagi
These reactions can be made more or less selective by fine-tuning the electric potentials, in addition to being ecologically benign. Their reliance on a power supply, however, limits their application in unpowered environments such as aerospace and deep sea.
A team of academics lead by Prof. Shinsuke Inagi from the Tokyo Institute of Technology (Tokyo Tech) in Japan revealed a solution to this self-contradictory challenge. The researchers demonstrated electrochemical polymerization of organic aromatic monomers without an external power supply in a recent paper published in Communications Chemistry.
Prof. Inagi explains, “We have seen a huge leap in the development of electrochemical reactors for carrying out organic synthesis, but most of them require a power source. We wanted to build a power-independent system to make the process more accessible. And we found the answer to our quest in streaming potential-driven electrochemistry.”
What exactly is this streaming potential that Prof. Inagi talks of?
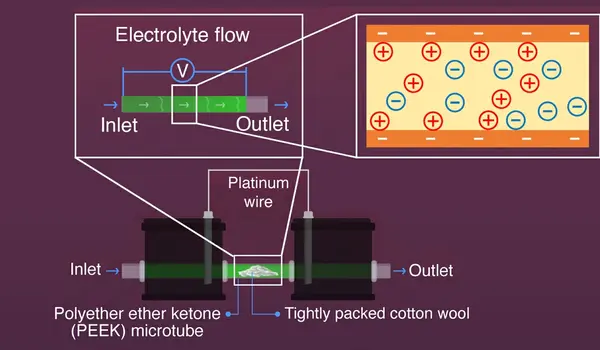
A pressure difference is formed when an electrolyte passes through a microchannel. This causes a charge imbalance, which gives rise to a streaming potential. For their studies, the researchers employed a bespoke two-chambered polyether ether ketone (or PEEK) cell connected by platinum wires and a PEEK microtube. To induce a pressure drop, this PEEK microtube was tightly packed with cotton wool. When scientists passed an electrolyte through the microtube, it generated a streaming potential that may give enough energy to drive the appropriate chemical reactions.
When the cell was activated, the electrodes in the two chambers experienced both upstream and downstream streaming potential, allowing the cell to function like a split bipolar electrode (BPE). This BPE system, along with the generated streaming potential of 2-3 volts, was responsible for producing circumstances favorable for organic monomer redox reactions.
The researchers picked two aromatic organic compounds to evaluate the polymerization abilities of this setup: Pyrrole (Py) and 3,4-Ethylenedioxythiophene (EDOT). Both of these monomers were electropolymerized into polypyrrole (PPy) and poly-EDOT (PEDOT) without the use of any external power source!
This innovative pressure-driven, ecologically friendly, and power-supply independent reactor opens up new possibilities for electrifying synthesis operations. This research can also be used to create novel electrochemical reactors for the synthesis of important organic chemicals and polymers. “The entire globe is working to make important industrial processes more environmentally friendly and cleaner. Because organic synthesis is at the heart of many chemical businesses, we attempted to build an electrosynthesis process that uses little resources and contributes to long-term development goals” Prof. Inagi concludes.