Crop plant genetic modification began 30 years ago when it became possible to change the magnitude of endogenous gene expression and introduce new genetic information using newly emerging plant transformation and recombinant DNA technology. The benefits of this technology, such as insect pest resistance and herbicide resistance, have been transformative for agriculture.
A team from the United States Department of Energy’s Brookhaven National Laboratory (BNL) has bred a plant that produces more oil by manipulating the availability of sugar for oil synthesis. The team, led by BNL’s John Shanklin, achieved these results by mimicking stem cells in plants such as energycane and Miscanthus using leaves from the fast-growing plant Arabidopsis.
The research is part of a biosystems design project led by the University of Illinois called Renewable Oil Generated with Ultra-productive Energycane (ROGUE), which aims to engineer two of the most productive American biomass crops, energycane, and Miscanthus, to produce an abundant and sustainable supply of oil that can be used to make biodiesel, bio-jet fuel, and bio-products.
Increasing the availability of sugar for oil synthesis. Researchers have bred a plant that produces more oil by manipulating the availability of sugar for oil synthesis.
The current project, “Mobilizing vacuolar sugar increases vegetative triacylglycerol accumulation,” expands on previous work published by the Shanklin group in 2017. This research demonstrated that impairing sugar export from leaves while blocking starch synthesis diverts sugars produced by photosynthesis towards fatty acid and oil synthesis.
“The novel aspect of this work was to minimize sugar accumulation in a large cellular storage compartment called the vacuole,” said BNL research associate Sanket Anaokar. “Our strategy was to prevent sugar from entering the vacuole and maximize its export. When these genetic modifications were applied to plants that are also inhibited in starch synthesis, the extra sugar was channeled into oil by the cell.”
Anaokar went on to say that one unexpected benefit of the group’s approach was that some of the remobilized sugar reduced the growth delays that are typically seen when the amount of exported sugar from the leaves and starches is reduced. The group will share what they’ve learned from their work with Arabidopsis with other ROGUE researchers, accelerating the innovation cycle.
However, metabolic engineering has been more difficult because metabolism is a common and highly regulated process. Any change in metabolism, for example, has the potential to alter carbon and energy sensing and signaling, a tightly regulated process analogous to blood sugar regulation in humans. It can also be argued that any attempt to engineer overall productivity through photosynthesis must engage with this regulatory system because carbon fixation is regulated by carbon and energy homeostatic regulatory systems.
Photosynthates, such as glucose, sucrose, and some of their derivatives, serve as both metabolic intermediates and signaling molecules in plant cells. These sugars serve as a starting point for the biosynthesis of fatty acids (FA). However, in comparison to the well-defined examples of sugar signaling in starch and anthocyanin synthesis, little was known about the role of signaling in regulating FA and lipid biosynthesis until recently.
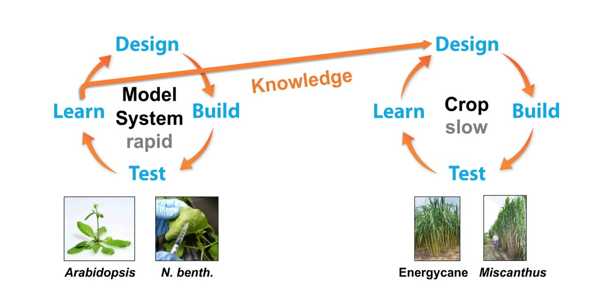
“Making multiple gene manipulations in energycane is far more difficult and time consuming, whereas with Arabidopsis we can rapidly develop and test different genetic and molecular biology modifications to identify the most effective combinations,” said Shanklin, BNL Biology Department Chair and ROGUE researcher. “Once we validate an approach using our model system, we can transfer that knowledge to other ROGUE researchers to use in slower-growing biomass crop plants.”
Sugars in a developing seed’s endosperm can play a variety of roles, including supplying carbon to the developing embryo and controlling gene expression. Because of the importance of seed size, sugar metabolism in seeds has an obvious agricultural context, but it also has a developmental context. It could be an example of a self-regulating, stable cycle of control and regulation.
Shanklin’s research is just one example of how ROGUE is working to increase the availability of sustainable biofuels while decreasing the use of petrochemicals. “This proof of concept in Arabidopsis now shows us that it is well worth moving into energycane and Miscanthus as a key step in making these viable sources of large amounts of oil for conversion into biodiesel and biojet fuel,” said ROGUE Director Stephen Long, Ikenberry Endowed University Chair of Crop Sciences and Plant Biology at Illinois’ Carl R. Woese Institute for Genomic Biolog.” The findings of this study have been published in Frontiers in Plant Science.