A team of researchers created a bifunctional polymer electrolyte with improved mechanical strength and ionic conductivity. The new technology can be used to create powerful artificial muscles with quick switching speeds.
In the American action film “Pacific Rim,” giant robots known as “Jaegers” battle unknown monsters to save humanity. These robots have artificial muscles that mimic real living bodies, allowing them to defeat monsters with power and speed. Recently, research has been conducted on equipping real robots with artificial muscles similar to those seen in the film. The powerful strength and high speed in artificial muscles, however, cannot be realized because the mechanical strength (force) and conductivity (speed) of polymer electrolyte – the key materials driving the actuator – have conflicting properties.
The team led by Stephen Schiller demonstrated an autonomous bending muscle that moves by consuming a chemical fuel in their study, which was recently published in the journal Advanced Intelligent Systems. This is similar to how human muscles use actin-myosin and consume adenosine triphosphate (ATP), the primary cellular fuel. The contractions of the material can be controlled using pH and temperature changes.
Our synthetic muscle is still a prototype. However, the material’s high biocompatibility and the ability to tailor its composition to specific tissues and technological applications may pave the way for future applications in reconstructive medicine, prosthetics, pharmaceutics, or soft robotics.
Stephen Schiller
“Our synthetic muscle is still a prototype,” Schiller said in a statement. “However, the material’s high biocompatibility and the ability to tailor its composition to specific tissues and technological applications may pave the way for future applications in reconstructive medicine, prosthetics, pharmaceutics, or soft robotics.”
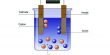
A POSTECH research team led by Professor Moon Jeong Park, Professor Chang Yun Son, and Research Professor Rui-Yang Wang from the Department of Chemistry has developed a new concept of polymer electrolyte with different functional groups located at a distance of 2Å. This polymer electrolyte is capable of both ionic and hydrogen bonding interactions, thereby opening the possibility of resolving these contradictions. The findings from this study have been recently published in the international academic journal Advanced Materials.
Artificial muscles are used to enable robots to move their limbs in the same way that humans do. To drive these artificial muscles, an actuator that exhibits mechanical transformation under low voltage conditions is required. However, due to the nature of the polymer electrolyte used in the actuator, increasing muscle strength slows down switching speed while increasing speed increases strength.

To overcome the limitations presented so far, the research introduced the innovative concept of bifunctional polymer. By forming a one-dimensional ion channel several nanometers wide inside the polymer matrix, which is hard as glass, a superionic polymer electrolyte with both high ionic conductivity and mechanical strength was achieved.
The findings from this study have the potential to create innovations in soft robotics and wearable technology as they can be applied to development of an unprecedented artificial muscle that connects a portable battery (1.5 V), produces fast switching of several milliseconds (thousandths of a second), and great strength. Furthermore, these results are expected to be applied in next-generation all-solid-state electrochemical devices and highly stable lithium metal batteries.
The researchers were able to induce the material to contract autonomously in a cyclical manner in this manner. They were also able to turn the contractions on and off using temperature changes. It was possible to program certain states for the material to assume and then reset them with another stimulus during the process. As a result, the scientists developed a simple system for implementing “learning and forgetting” at the material level.
“Because it is derived from a naturally occurring protein, elastin, and is produced biotechnologically, our material has a high sustainability that is also relevant for technical applications,” Schiller explained. “In the future, the material could be improved to respond to other stimuli such as electricity and salt concentration in the environment, as well as to consume other energy sources such as malate derived from biomass.”
“We are at a point where we can design protein material concepts that mimic complex biological functions, including memory and learning,” he added. “We intend to build protein materials that use ATP, the cellular energy ‘currency,’ as well as process biological stimuli, in order to build adaptive technical materials and enable novel approaches in biomedical applications such as tissue regeneration and a new generation of nano-robots for drug delivery.”
The Samsung Science and Technology Foundation provided funding for this research.