The main features of Rutherford’s Nuclear model of the atom are:
i) In an atom, the entire mass and positive charge are concentrated in a very small region at the center known as a nucleus. It was observed that the radius of the nucleus is of the order of 10-13cm whereas the radius of the atom is the order of 10-8 cm. This means the size of a nucleus is extremely small (only about 1/100000) as compared to the size of an atom.
ii) The magnitude of the positive charge in the nucleus (number of protons) is different for different atoms.
iii) The electrons are not stationary but are revolving around the nucleus at speed like planets revolving around the Sun. As a result, the electrons are called planetary electrons.
iv) Most of the space in an atom between the nucleus and the revolving electron is empty.
Limitations of Rutherford’s Atomic model
The Rutherford’s model for the structure of atoms has several flaws.
1) This model was made analogous to the solar system where the nucleus may be compared to the sun, and the electron to the planets In space the planets are neutral and attract each other according to the law of gravitation. However, the electrons orbiting the nucleus are negatively charged and these electrons repel each other.
2) It was shown by Clark Maxwell that a charged body moving under the influence of attractive forces loses energy continuously in the form of electromagnetic radiation. Thus, unlike a planet, the electron is a charged body and it should emit radiations while revolving around the nucleus. As a result, the electron should lose energy at every turn and move closer and closer to the nucleus following a spiral path. Consequently, the orbit will become smaller and smaller and finally the electron will fall into the nucleus. In other words, the atom should collapse. However, this never happens. Therefore we expect some drawbacks. in the atomic model.
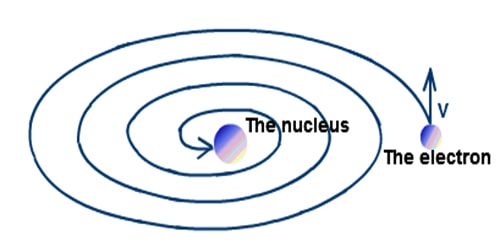
Fig: Continuous emission of energy as a result electron falls to the nucleus.
3) Rutherford model also failed to explain the existence of certain definite lines in the hydrogen spectrum. If the electrons were to lose energy continuously as discussed above the atomic spectrum of hydrogen should have been continuous. However, the spectrum of hydrogen has been found to be discontinuous in the form of characteristic lines of definite wavelengths.
4) The model does not give any idea of the shape and size of the orbits of electrons. for atoms having more than one electron, the model failed to explain the manner in which these electrons revolve around the nucleus.











