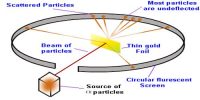
Limitation of Rutherford’s Atom Model
Although Rutherford’s atom model is more logical than Thomson’s model, it has some defects or limitations. The limitations of this model are discussed below:
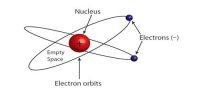
According to electromagnetic theory when a charged particle is in motion with acceleration, then that particle continues to radiate energy in the form of electromagnetic waves and thereby its velocity is decreased. Since the electron revolving in its orbit is acted upon by the centripetal force its motion is accelerated. Thus it will lose energy by the emission of electromagnetic radiation. So due to the attractive force of the nucleus, it will move in orbits of continually decreasing radii and its path will be spiral as shown in the figure. Finally, it will fall upon the nucleus itself and will disappear. So, in this model, there could not be any atom with the positively and negatively charged parts existing separately in it.
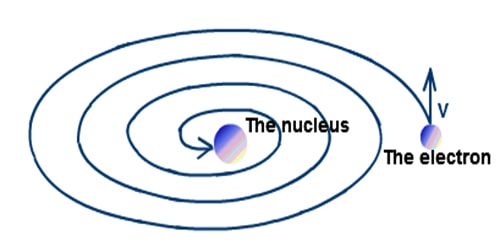
Fig: Continuous emission of energy as a result electron falls to the nucleus.
According to Rutherford’s model, electrons while rotating around the nucleus in an orbit will radiate electromagnetic waves of all wavelengths and will demonstrate continuous spectra. But in the case of hydrogen line spectrum twin of a fixed wavelength is obtained. It has been found from the calculation that an electron rotating in an orbit will exhaust all of its energy within 108 seconds and will tall upon the nucleus. So, the atom of this type cannot have permanent existence. This as also contrary to the experimental result. Experimentally it is found that atoms, like hydrogen atoms, emit line spectra of fixed wavelengths only and not of all wavelength.
Hence, there is no conformity of the experimental results with Rutherford’s model. So Rutherford’s model is not acceptable.