Age-related mitochondrial alterations are linked to a reduction in mitochondrial function. Because of mutations and oxidative damage caused by reactive oxygen species, mitochondrial DNA volume, integrity, and functionality decline with age (ROS).
A typical aspect of many age-related disorders is that the mitochondria of cells begin to fail. While the origin is unknown, Buck Institute scientists have uncovered a novel process by which mitochondria begin to malfunction, which offers up new avenues for researchers to investigate how to begin to solve the problem.
“Now that we have this different hypothesis of why mitochondrial dysfunction occurs during aging and age-related diseases, we open up a completely different way to think about, measure, mitigate, and reverse that process,” said Buck Fellow Chuankai (Kai) Zhou, Ph.D., senior author of a study published in the issue of eLife, a leading journal on biomedical research established by the Howard Hughes Medical Institute, Max Planck Society, and Wellcome Trust.
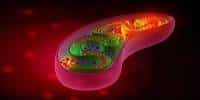
Mitochondria are widely referred to as the “powerhouse” of the cell since the bean-shaped structures provide the energy units that every cell requires to function. They are engaged in a variety of key functions, including immune response, inflammation, and metabolism.
Our discovery that the molecule Tom70 is coordinating both aspects crosses two independent fields of study. Now that we have this different hypothesis of why mitochondrial dysfunction occurs during aging and age-related diseases, we open up a completely different way to think about, measure, mitigate, and reverse that process.
Chuankai (Kai) Zhou
Dysfunction of the mitochondria is implicated in most chronic diseases, said Zhou, including neurodegenerative disorders, cardiovascular diseases, cancer, diabetes, and obesity. A growing body of evidence indicates that this dysfunction contributes to aging in general.
Zhou’s team aimed to investigate the role of “mitochondrial biogenesis” in mitochondrial dysfunction throughout aging. Mitochondrial biogenesis is the biological process that results in the formation of new mitochondria. It regulates the quantity and quality of mitochondria in cells, both of which deteriorate with age. Scientists have already described two major steps in mitochondrial biogenesis over the last few decades: the transcriptional activation of mitochondrial protein in the cellular nucleus in response to nutrient/metabolic signals, and the import of these newly synthesized mitochondrial proteins from the cytosol into mitochondria.
However, it was unknown how these two processes of mitochondrial biogenesis work together to speed up the synthesis and import of mitochondrial proteins. Zhou’s team discovered that Tom70, a conserved receptor molecule on the surface of mitochondria, may be in charge of coordinating these two phases by controlling the transcriptional activity of mitochondrial proteins inside the nucleus.

Tom70 has previously been shown to aid in the import of newly produced mitochondrial proteins. However, Zhou pointed out that the amount of protein produced must be fine-tuned to match the needs and import capability of the mitochondria while not overshooting production to the point where excess accumulates within the cell, causing harm and death.
In the current study, his team employed yeast as a model to see if Tom70 governs mitochondrial biogenesis by managing the intricate balance between the creation of proteins intended for the mitochondria and the rate at which these proteins may be escorted into the mitochondria. They discovered it did. Fruit flies have a similar regulatory function as Tom70.
The two stages of communication concerning requirements and uptake into the mitochondria have not before been linked. “Our discovery that the molecule Tom70 is coordinating both aspects crosses two independent fields of study,” Zhou said.
Surprisingly, they discovered that Tom70 diminishes with age, which is related with decreased mitochondrial biogenesis. Tom70 decreases in old fruit flies and rats have previously been documented. The researchers went on to show that boosting the level of Tom70 in yeast postponed mitochondrial dysfunction and improved lifespan.
This discovery is consistent with what has been known for more than a decade: boosting Tom70 can protect human cells against various chronic diseases, such as cardiovascular disease, but the molecular specifics of how this occurs were unknown. “We uncovered a new role for this protein and propose a mechanism for how Tom70 can do good for the cell,” Zhou added.
Zhou believes that this insight can point to new possibilities in aging research in a variety of ways. The Tom70 pathway, for example, has been connected to the increased lifespan found with calorie restriction. “Furthermore, if our idea is correct, it will inevitably lead to products such as supplements or medications. It simply takes time “he said.
Zhou hopes to delve deeper into yeast genetics to learn more about the mechanism, such as how Tom70 communicates with the nucleus. “That’s the beauty of basic science; it gives you something to investigate that didn’t exist before,” he explained. “We are excited about translation, but we are even more excited about something brand new, that no one knew about before.”