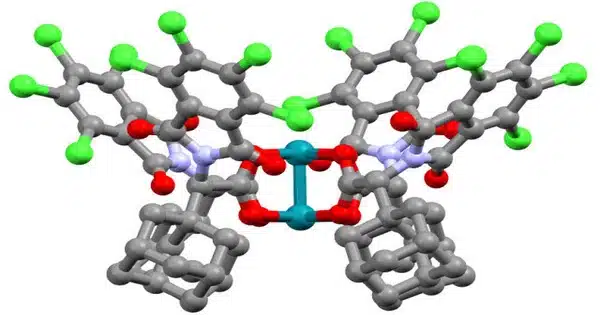
Biosynthesis of drug precursors frequently involves complex chemical events and processes. Carrier-mediated ring formation is important in the production of pharmacological precursors in some circumstances. The whole actinopyridazone biosynthetic pathway has been revealed, demonstrating that an unprecedented carrier protein-mediated ring-formation phase is critical to its synthesis.
Nitrogen-nitrogen bond-containing cyclic molecules such as pyrazole, triazole, pyridazine, and many more are essential building blocks for a wide range of natural and manufactured therapeutic chemicals. Some of these chemicals’ production is dependent on the creation of nitrogen-nitrogen (N-N) single bonds between amino acids. However, the mechanisms that allow for such a wide range of chemicals are poorly understood.
Dr. Kenichi Matsuda and Professor Toshiyuki Wakimoto at Hokkaido University led a team to elucidate the biosynthetic pathway of actinopyridazinone, an N-N bond-containing cyclic compound that is an important scaffold for synthetic drugs. Their findings were published in the journal Angewandte Chemie International Edition.
Actinopyridazinone is produced by Streptomyces, a genus of bacteria that is the source of the vast majority of antibiotics of natural origin. It is the first naturally occurring compound with a dihydropyridazinone ring.
Professor Toshiyuki Wakimoto
“Actinopyridazinone is produced by Streptomyces, a genus of bacteria that is the source of the vast majority of antibiotics of natural origin,” Wakimoto says. “It is the first naturally occurring compound with a dihydropyridazinone ring. This ring is also regarded as a ‘wonder nucleus,’ because it has been intensively explored as a pharmacological precursor.”
Previously, the researchers employed bioinformatics to uncover a collection of gene sequences that are likely involved in the production of natural products containing N-N bonds, and from these genome sequences, they discovered a unique class of molecules known as actinopyradizones. With a series of genetic and biochemical experiments, they were also able to unveil the first steps in the pathway; in this study, they focused on understanding how the dihydropyridazone ring is formed.
The gene cluster apy is the biosynthetic gene cluster associated with actinopyradizone synthesis. It contains 17 potential genes; knockout studies indicated that ten of these – apy1, apy2, apy3, apy4, apy6, apy8, apy9, apy10, apy11 and apy13 – were necessary for actinopyradizone synthesis. Biochemical analyses of the knockouts allowed the team to deduce that Apy3, an AMP-dependent synthetase/ligase, Apy4, a serine hydrolase, and Apy6, a carrier protein-rhodanese fusion, were the key proteins responsible for the formation of the dihydropyridazone ring.
“Apy6 functions as a carrier molecule; and Apy3 loads the intermediate compound onto Apy6,” Matsuda elaborates. “Apy4 then catalyzes the removal of an acetyl group (-COCH3); the resulting molecule is unstable and spontaneously reacts to form a dihydropyridazone ring. The most notable feature of actinopyridazone biosynthesis is the unprecedented carrier protein-mediated machinery for dihydropyridazinone formation.”
Matsuda said that this study is the first description of the biosynthetic pathway for actinopyradizone, and is only the second study to report the enzyme-dependent biosynthesis of a N-N bond-containing ring structure. The first such compound is piperazic acid, whose biosynthetic pathways are completely unrelated; hence, this study has also highlighted that the biosynthetic pathways of N-N bond-containing cyclic compounds are very diverse.