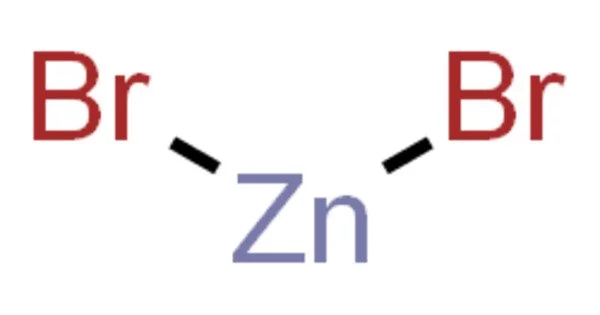
Zinc bromide is a chemical compound with the formula ZnBr2. It is a hygroscopic compound that dissolves easily in water. It is a colorless salt with many properties in common with zinc chloride (ZnCl2), including high solubility in water, which forms acidic solutions, and good solubility in organic solvents. It is a chemical component that is not flammable. It is hygroscopic and reacts with water to form the dihydrate ZnBr2•2H2O.
Zinc bromide is primarily used in the oil industry, electrolysis, catalysis, the electrical industry, and metalworking. It is also used as a radiation shielding transparent film.
Properties
Zinc Bromide is an inorganic, non-flammable compound which is used as Lewis acid in organic chemistry. It is a hygroscopic compound that is easily soluble in water and forms dihydrate as ZnBr2. 2H2O. It is a colourless salt which is soluble in water to form acidic solutions.
- Chemical formula: ZnBr2
- Molar mass: 225.198 g/mol
- Appearance: white crystalline powder, hygroscopic
- Density: 4.20 g/cm3 (20 °C); 4.22 g/cm3 (25 °C)
- Melting point: 394 °C (741 °F; 667 K)
- Boiling point: 697 °C (1,287 °F; 970 K)
- Solubility in water: 388 g/100 mL (0 °C); 675 g/100 mL (100 °C, for the anhydrous material)
- Solubility: very soluble in alcohol, ether, acetone, tetrahydrofuran

Production
ZnBr2 · 2H2O is prepared by treating zinc oxide or zinc metal with hydrobromic acid.
ZnO + 2 HBr + H2O → ZnBr2·2H2O
Zn + 2 HBr → ZnBr2 + H2
Anhydrous material can be created by dehydrating the dihydrate with hot CO2 or by reacting zinc metal and bromine. The anhydrous derivative is also obtained by sublimation in a stream of hydrogen bromide.
Uses
Zinc bromide is used in the following applications:
- In organic chemistry as a Lewis acid.
- It is the electrolyte in the zinc bromide battery.
- In oil and natural gas wells, Solutions containing zinc bromide (ZnBr2) are used to displace drilling mud when transitioning from the drilling phase to the completion phase.
- It is used as a transparent film shield against radiation.
Zinc bromide solutions can be used as a transparent radiation shield. To be used as a window on a hot cell, the space between two glass panes is filled with a strong aqueous solution of zinc bromide with a very high density. This type of window has an advantage over lead glass in that it does not darken as a result of radiation exposure.
Safety
The safety concerns are similar to those for zinc chloride, which has a toxic dose for humans of 3-5 g. It is a corrosive substance that is hazardous to the environment. Contact with the eyes and skin can result in severe burns and blindness, so remove contaminated clothing immediately and thoroughly rinse the affected area with water.