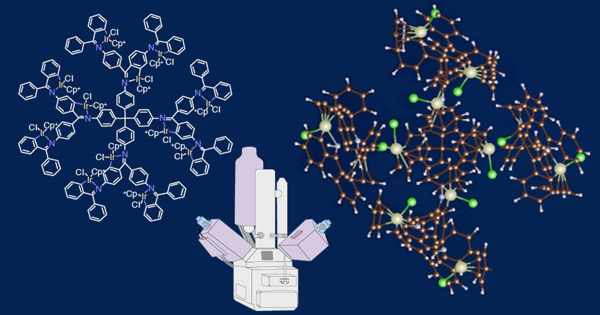
A complex is a substance that contains a metal atom or ion as well as a group of neutral molecules or anions known as ligands. Coordination compounds are neutral substances that contain at least one ion as a complex. A coordination complex is the result of a Lewis acid-base reaction in which neutral molecules or anions (called ligands) form coordinate covalent bonds with a central metal atom. Ligands are Lewis bases, which means they have at least one pair of electrons to donate to a metal atom/ion.
Scientists have demonstrated that high-angle annular dark-field scanning transmission electron microscopy can be used to determine complex conformational structures of both crystalline and amorphous polynuclear non-planar coordination molecules. They were successful in determining the different conformations of a highly branched coordination compound molecule using iridium as a tracer metal. This has enabled the imaging and design of complex inorganic and organic molecules.
Coordination compounds have molecular structures that include one or more metal atoms in the center surrounded by nonmetal atoms. Their intriguing physical and chemical properties, which have important applications in material science, are heavily influenced by their molecular structure. As a result, not only is a definitive analysis of their molecular structure required for understanding their properties, but also for designing specific coordination compounds with targeted functions.
High-angle annular dark-field scanning transmission electron microscopy can be used to determine complex conformational structures of both crystalline and amorphous polynuclear non-planar coordination molecules, scientists have shown.
Although several analytical methods for determining the structural properties of coordination compounds are available, each has its own set of limitations. X-ray crystallography, for example, can only determine the structure of crystalline compounds, whereas nuclear magnetic resonance cannot provide accurate results when paramagnetic atoms are involved.
A more recent microscopy technique, high-angle annular dark-field scanning transmission electron microscopy, which has revolutionized molecular imaging by allowing real-time visualization of single coordination molecules, is also restricted to simple and planar molecules. Hence, the structural determination of various conformations of both crystalline and amorphous polynuclear coordination molecules remains unexplored.
To close this gap, a team of Tokyo Institute of Technology researchers led by Professor Kimihisa Yamamoto and Associate Professor Takane Imaoka developed a novel imaging method in HAADF-STEM using a metal-atom tracer to determine the conformational structures of complex and highly branched polynuclear coordination compounds.
Their findings have been published in the journal Science Advances. Prof. Imaoka explains the new method, “We synthesized iridium fixated dendritic phenylazomethine (DPA) compounds using iridium as a metal tracer because its high atomic number (Z=77) will provide better visualization with HAADF-STEM.” Then, we determined the optimum operating conditions for HAADF-STEM, under which the different conformations of these highly branched DPA compounds could be determined with the highest accuracy.”
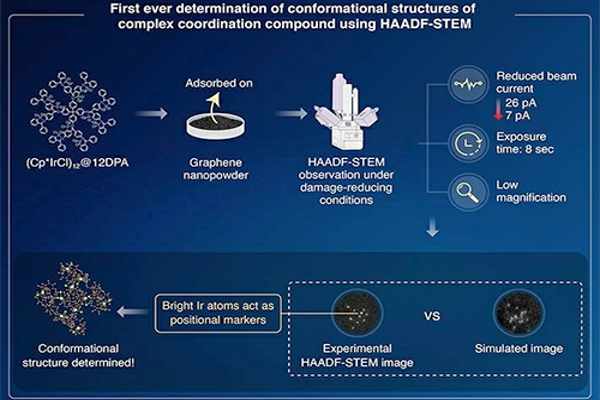
The researchers observed samples of iridium-DPA compound dispersed on the surface of graphene nanopowder under a variety of operating conditions to determine the optimum operating conditions for HAADF-STEM. They discovered that lowering the beam current to 7 pA, the exposure time per pixel to 8 microseconds, and using low magnification helped reduce damage to the iridium-DPA compound and allowed them to successfully observe its structure. In the HAADF-STEM images, the iridium atoms appear as bright spots, indicating their position in the molecule’s structure.
After obtaining the HAADF-STEM image of the iridium-DPA molecule under optimal conditions, the researchers compared it to simulated images of all possible molecule conformations to find the closest match. The structures captured in the experimental HAADF-STEM images match the simulated conformational structures extremely well. Thus, by comparing HAADF-STEM and simulated images, the most accurate conformational orientation of a molecule can be easily determined.
This heavy metal-guided HAADF-STEM technology’s potential applications are not limited to structural analysis coordination compounds. Prof. Imaoka highlights future work by saying, “Our research is a game-changer in imaging the conformational structures of complex macromolecules. Because this technology works for both crystalline and amorphous compounds, we believe it can be used to determine the structures of multinuclear peptides via complexation with tracer metal atoms, and research in this area is already underway.”