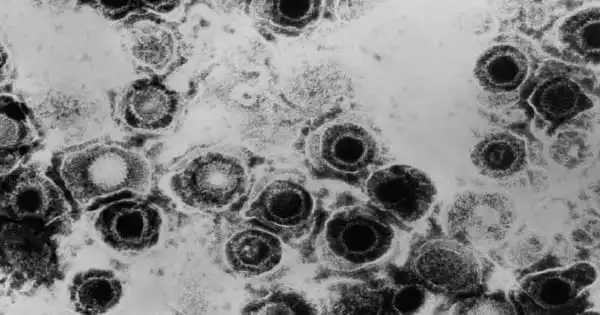
Herpesvirus, any virus in the Herpesviridae family. These viruses are pathogenic (cause disease) in a wide range of animals, including humans, monkeys, birds, frogs, and fish. Human herpesviruses, such as herpes simplex virus type 1 (HSV-1) and herpes simplex virus type 2 (HSV-2), are among the best-studied (HSV-2). Unlike HSV-1, which is transmitted orally and causes cold sores and fever blisters around the mouth, HSV-2 is transmitted sexually and is the primary cause of the condition known as genital herpes.
A new study published in mBio, an open access journal of the American Society for Microbiology, has paved the way for a novel approach to fighting herpesviruses. The study found that targeting two metal ion-dependent enzymes of human herpesviruses with two compounds, AK-157 and AK-166, can inhibit virus replication. The discovery opens up new avenues for the development of anti-herpesvirus agents.
Researchers from the University of Connecticut School of Medicine and School of Pharmacy have published an article in mBio titled “Two-Metal Ion-Dependent Enzymes as Potential Antiviral Targets in Human Herpesviruses,” which could pave the way for a novel approach to attacking herpesviruses. The study demonstrated that two compounds, AK-157 and AK-166, can inhibit virus replication by targeting two-metal ion-dependent enzymes of human herpesviruses. According to the researchers, the discovery opens up new avenues for developing anti-herpesvirus agents.
The majority of herpesvirus drug discovery efforts have focused on nucleoside analogs that target viral DNA polymerases. We are pursuing a strategy that focuses on two-metal-ion-dependent viral enzymes
Sandra K. Weller
“Many people are familiar with herpes simplex viruses, but there is a family of 9 different herpesviruses, including cytomegalovirus (CMV), which causes significant problems in immunocompromised people, transplant recipients, and chemotherapy patients, for example. We need better therapeutic agents that can be used in these extremely vulnerable populations” Dennis Wright, Ph.D., a professor of medicinal chemistry in the University of Connecticut’s School of Pharmacy, was one of the study’s co-authors.
“Right now, the therapeutic agents that are available aren’t particularly effective in terms of treating all viruses, and many of them have significant dose-limiting toxicities and associated side effects.”
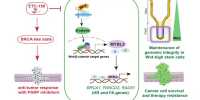
Ideally, Wright said, there would be a single drug that would prevent the reactivation of all nine herpesviruses. Sandra K. Weller, Ph.D., a distinguished professor of molecular biology and biophysics in the University of Connecticut School of Medicine, identified targets that would allow this. She discovered herpesvirus enzymes that require two magnesiums in order for the virus to replicate. “The majority of herpesvirus drug discovery efforts have focused on nucleoside analogs that target viral DNA polymerases. We are pursuing a strategy that focuses on two-metal-ion-dependent viral enzymes” Weller stated.
The researchers tested the ability of a panel of compounds to inhibit specific 2 metal ion-dependent enzymes as well as herpesvirus replication in test tube studies. HIV integrase inhibitors, the anti-influenza agent baloxavir, three natural products previously shown to have anti-herpes simplex virus (HSV) activity, and two 8-hydroxyquinolones, AK-157 and AK-166, were among the compounds tested.
“This enzyme family consists of structurally related proteins with shared active sites that contain conserved carboxylates predicted to coordinate divalent cations required for catalysis.” TMID enzyme-targeting compounds, such as HIV integrase and influenza endoribonuclease, have been developed for clinical use. The researchers write, “HIV integrase inhibitors have been reported to inhibit replication of herpes simplex virus (HSV) and other herpesviruses; however, the molecular targets of their antiviral activities have not been identified.”
While HIV integrase inhibitors have been shown to inhibit herpesvirus replication, the researchers discovered that the integrase inhibitors had weak overall anti-HSV-1 activity. However, the researchers discovered that 8-hydroxyquinolones had potent antiviral activity against both HSV-1 and CMV, as well as the ability to inhibit one or more of the two metal ion dependent enzymes. This opens the door to the development of dual-targeting agents against herpesviruses.
“The integrase inhibitors had weak overall anti-HSV-1 activity, whereas the 8-HQs inhibited both HSV-1 and cytomegalovirus” (CMV). Contrary to previous reports, the target-based analysis revealed that none of the antiviral compounds inhibited ICP8. Baloxavir, on the other hand, inhibited the HSV polymerase proofreading exonuclease, whereas AK-157 and AK-166 inhibited the alkaline exonuclease UL12. Furthermore, AK-157 inhibited the catalytic activity of the HSV polymerase, suggesting the possibility of developing dual-targeting agents against herpesviruses.”