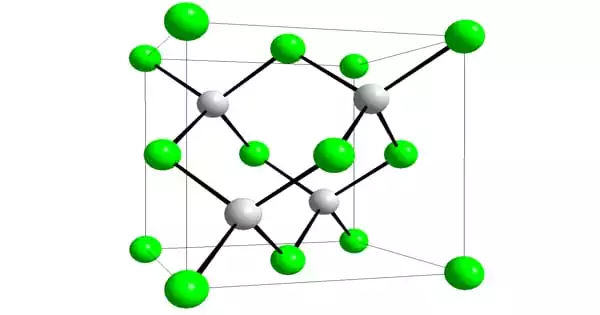
Aluminium antimonide (AlSb) is a semiconductor combining aluminium and antimony from the group III-V family. Aluminum arsenic antimonide, aluminum gallium antimonide, and aluminum indium antimonide can be synthesized by alloying it with other III-V semiconductor materials. 0.61 nm is the lattice constant. At 300 K, the indirect band gap is roughly 1.6 eV, while the direct band gap is 2.22 eV. Because of its proclivity to generate (Sb3-) ions, it is extremely flammable.
At 300 K, its electron mobility is 200 cm2•V-1•s-1 and its hole mobility is 400 cm2•V1•s1. It has a refractive index of 3.3 at 2 μm wavelength and a dielectric constant of 10.9 at microwave frequencies.
Aluminium antimonide is a chemical compound formed by the elements aluminum and antimony. It’s employed as a semiconductor as well as in photonics. AlSb can react with other III-V elements to form ternary compounds such as AlInSb, AlGaSb, and AlAsSb.
Properties
Aluminium antimonide is a solid that is black in color. It is a type of semiconductor. It combines with acids to form stibine. It functions as a reducing agent. It possesses the qualities of both a salt and an alloy. Because of the reducing tendency of the antimonide (Sb3-) ion, it is quite flammable. When it burns, it emits aluminum oxide and antimony trioxide.
- Molecular Weight: 148.73
- Appearance: Black crystals
- Melting Point: 1060 °C
- Boiling Point: 2467 °C
- Density: 4.26 g/cm3
- Solubility in H2O: N/A
Applications
A chemical compound is aluminium antimonide. It has the chemical formula AlSb. It contains aluminum and antimonide ions. Compounds similar to it, such as gallium antimonide and indium antimonide, can be used to make LEDs and infrared detectors, however aluminium antimonide does not perform as well. Instead, it is utilized to improve the performance of the other compounds by preventing faults from occurring throughout the manufacturing process.
Aluminum antimonide finds applications in the following:
- Electro-optical devices
- X-ray and Gamma-ray radiation detection.