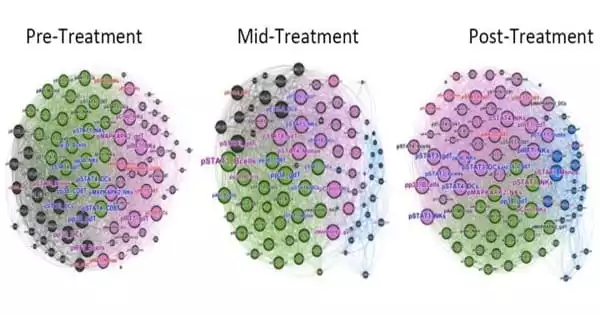
According to research, chronic viral infections have a profound and long-lasting impact on the immune system in ways similar to those seen during aging. Researchers used systems immunology and artificial intelligence to profile and compare immune responses in an aging population, people with HIV on anti-retroviral therapy and people infected with hepatitis C before and after the virus was treated with a drug with a 97 percent cure rate.
Chronic viral infections, according to research from the Buck Institute and Stanford University, have a profound and long-lasting impact on the human immune system in ways that are similar to those seen during aging. The findings have been published in the Proceedings of the National Academy of Sciences.
Researchers used systems immunology and artificial intelligence to profile and compare immune responses in an aging population, people with HIV on long-term anti-retroviral therapy and people infected with hepatitis C (HCV) before and after the virus was treated with a drug that has a 97 percent cure rate. T cell memory inflation, upregulation of intracellular signaling pathways of inflammation, and decreased sensitivity to cytokines in lymphocytes and myeloid cells are all shared immune system changes.
Chronic inflammation caused by immune system dysfunction is linked to many aging diseases. The question of whether chronic viral infection contributes to age-related immune dysfunction remains unanswered, but studies like this one provide a starting point for finding out. At this point, it is clear that both aging and chronic viral infections have profound and permanent effects on immunity.
David Furman
“Chronic inflammation caused by immune system dysfunction is linked to many aging diseases,” says David Furman, Ph.D., Buck Institute associate professor and senior author of the study. “The question of whether chronic viral infection contributes to age-related immune dysfunction remains unanswered, but studies like this one provide a starting point for finding out. At this point, it is clear that both aging and chronic viral infections have profound and permanent effects on immunity.”
The difference between acute and chronic viral infections
In the case of acute viral infections, the body is usually able to clear the offending agent, and the immune system (in the best-case scenario) produces antibodies that protect against similar infections — think of common colds and seasonal flus. But, in addition to HIV and HCV, there are viruses that can survive in the body and establish “host-parasite housekeeping,” in some cases without the host’s knowledge. According to Furman, 70 to 90 percent of the population is infected with cytomegalovirus, which is harmless in healthy people but dangerous in pregnant women or those with compromised immune systems. Chronic infections can also be caused by various herpes viruses (which cause genital herpes, cold sores, chicken pox/shingles, and mononucleosis).
“Each of us has our own virome; it’s a collection of viral infections you’ve had throughout your life,” Furman explains. “You were most likely infected by 12 or 15, if not more, viruses that you were unaware of. Fortunately, technology now allows us to profile these infections in the human population, which is assisting us in moving these types of investigations forward.” According to Furman, this is the first study to fully incorporate the concept of systems immunology and holistically analyze the immune system across different patient cohorts using the same technological platforms.
Reason for hope — results show some plasticity in immune response
Despite being treated with virus-suppressing drugs for over ten years, patients with HIV had immune system dysregulations, according to the study. However, HCV virus clearance (via the drug sofosbuvir) restored some cellular sensitivity to interferon-a, which inhibits viral replication. “This plasticity means that there is room for intervention in both chronic viral infections and aging,” Furman says. “It’s simply a matter of identifying and comprehending the molecular pathways and networks at work.” STAT1, the primary transcription factor activated by interferons, was found to be altered in this study. STAT1 is essential for normal immune responses, particularly against viral, mycobacterial, and fungal pathogens.
Implications for COVID-19?
According to Furman, we are in the midst of a “living” experiment with the COVID-19 pandemic. Future research is needed to determine whether the immune system’s functional imprinting is hardwired to only involve the chronic nature of specific infections, or whether relatively short-lived but vigorous inflammations like COVID-19 also leave a long-lasting imprint on the immune system. “Has the immune system of those infected with the coronavirus been severely compromised?
That’s just a theory; we don’t know what will happen “Furman is working on projects involving COVID-19 and immunity with Stanford University and the University of California, San Francisco.