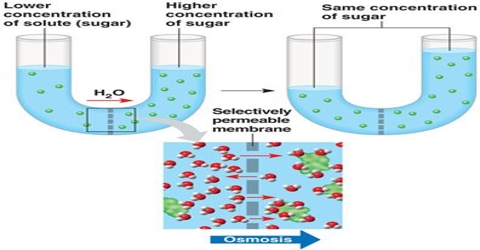
Diffusion pressure deficit: Diffusion pressure expresses the potential aptitude to diffuse from an area of the greatest concentration to an area of lesser concentration. Diffusion pressure deficit is the reduction in diffusion pressure of the solvent in a system over its pure state.
Important points of Diffusion pressure deficit:
- DPD was originally called suction pressure.
- It is measured in atmospheres.
- DPD is the difference between the diffusion pressure of a solution and pure solvent.
- DPD = OP – TP where OP = Osmotic pressure and TP = Turgor pressure.
- Water moves from lower DPD to higher DPD.
The cell-to-cell movement of water in plants, insofar as it is aggravated by osmotic and imbibitional mechanisms, can be effectively interpreted only in terms of the three physically distinct quantities of osmotic pressure (OP), turgor pressure (TP), and diffusion pressure deficit (DPD). No one of these three quantities alone adequately characterizes the water relations of a plant cell insofar as they can be interpreted on the basis of a diffusional mechanism. The magnitude of all three of them must be known for the complete evaluation of the water relations of a cell.