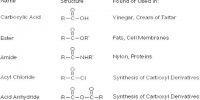
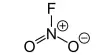
Preservation process of the chemicals:
(i) Benzoate: Sodium benzoate is used to preserve Jam, Jelly, carbonated beverage, fruit Juice and pickle. Its activity increases with the increasing of acidity. Dissolving in food it produce benzoic acid which is absorbed in the food cell As a result the pH value reduced less than two and hence micro organism cannot lead life and generate in that acidic medium. Hence they are destroyed.
(ii) Sorbate: It is very active to prevent east & mould and is less energetic to prevent bacteria. Bread, biscuits, cake, syrurn, fruit juice, Jam, Jelly, pickles are preserved by using sorbate. It is found in market as Calcium, Sodium or potassium benzoate. Sorbate create acidic environment where pH value is 4-5 and destroy microorganism. Sorbate absorbed in human body and produce CO2 and H2O after decomposition.
(iii) Acetic acid: Acetic acid is used as vinegar 6-10% acetic acid is exists in vinegar. It reduces the value of pH of foodstuffs It is used in a great deal to prepare pickle and sauce.