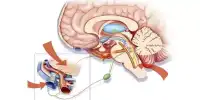
Opioid drugs continue to be a first-line treatment for severe pain, whether caused by surgery, a sudden injury, or nerve damage. However, as opioid addiction and overdose mortality in the United States hit new highs, the need for safer alternatives to treat acute pain has become more pressing. A new class of chemicals appears to be promising.
Scientists at Scripps Research in Florida have developed a new class of pain-relieving chemicals that, like morphine and other medications, deliver relief by activating opioid receptors, but without the many dangerous and undesired side effects that have contributed to opioid-related overdose and deaths.
In the journal Proceedings of the National Academy of Sciences, biochemist Laura Bohn, PhD, and colleagues describe SR-17018, a compound that activates the same pain-relieving receptor as opioid drugs such as morphine, oxycodone, and fentanyl; however, it binds to opioid receptors differently than those drugs, leaving the opioid receptor open and available to the body’s own natural pain-relieving substances, thereby apparently augmenting pain relief. The group demonstrated that the molecule performed particularly well in mice trials of chemotherapy-induced neuropathic pain in a study published earlier this year, the scientists wrote.
The authors of the current paper have made significant progress in understanding why these medications appear so dissimilar.
SR-17018 is the first biased agonist of the mu opioid receptor that does not cause tolerance with persistent usage. This is an appealing trait for possible application in the setting of chronic, severe pain.
Laura Bohn
“We show that these molecules bind to a different location on the receptor than conventional opioids. As a result, they appear to leave the receptor on while remaining sensitive to endogenous opioids “Bohn, who is the chair of the Scripps Research Department of Molecular Medicine in Jupiter, Florida, agrees. “In neuropathic pain, they are considerably superior to morphine and oxycodone; additionally, SR-17018 does not impair breathing.”
The authors also described a similar chemical that, because it is more effective, causes respiratory suppression, but only at larger levels than those required to reduce pain. Importantly for safety, when given at high enough concentrations to inhibit respiration, this chemical, SR-14968, demonstrated responsive to the overdose rescue medicine naloxone.
Perhaps most critically, for patients suffering from severe chronic pain, SR-17018 demonstrated the capacity to give long-term pain relief without tolerance, avoiding the problem of decreased efficacy over time, which necessitates higher doses, raising the risk of overdose.

The new compounds are referred to as “biased agonists” by the paper’s first author, Edward L. Stahl, because they activate the mu opioid receptor in a way that preferentially engages one of its signaling pathways, the one that provides pain relief, over other pathways, such as those that lead to suppressed breathing.
“SR-17018 is the first biased agonist of the mu opioid receptor that does not cause tolerance with persistent usage,” explains Stahl, a senior staff scientist in the Bohn lab. “This is an appealing trait for possible application in the setting of chronic, severe pain.”
He adds that the new compounds were designed to avoid the “beta-arrestin” signaling cascade that leads to opioids’ dangerous and unwanted characteristics, such as respiratory suppression, a cause of overdose, and constipation.
Opioid drugs continue to be a first-line treatment for severe pain, whether caused by surgery, a sudden injury, or nerve damage. However, as opioid addiction and overdose mortality in the United States hit new highs, the need for safer ways to treat acute pain has become more pressing, according to Bohn.
Bohn and her colleagues have established the viability of disentangling opioids’ pain-relieving capabilities from their undesirable side effects in research spanning more than two decades. Her research has not only increased understanding of how opioid receptors interact to control different physiological responses, but it has also pointed the science toward possibly safer methods for treating severe pain.
Bohn’s lab has created a number of chemicals that, in mouse studies, reduce beta-arrestin signaling activation. Other opioid-related issues, such as dependency or addiction, continue to pose difficulties. The fact that SR-17018 does not cause tolerance with long-term use is encouraging, according to Bohn. She also mentions how quickly it fades.
“The compound tapered nicely and slowly. This, in and of itself, may assist to alleviate some of the dependent issues. Morphine, for example, delivers a fast rush followed by a quick clear, and you need the rush again.”
Moving forward, the team will continue to improve and test the chemicals in order for them to be examined in a clinical setting. “Severe and persistent pain caused by surgery, nerve injury, or trauma necessitates significant pain medication,” explains Bohn. “More secure solutions are required. We believe that these novel chemicals represent a significant step forward.”
Cullen L. Schmid, Agnes Acevedo-Canabal, Cai Read, Travis Grim, Nicole Kennedy, and Thomas D. Bannister are the other co-authors of the publication, “G protein-signaling biased mu opioid receptor agonists that cause prolonged G protein activation are noncompetitive agonists.”