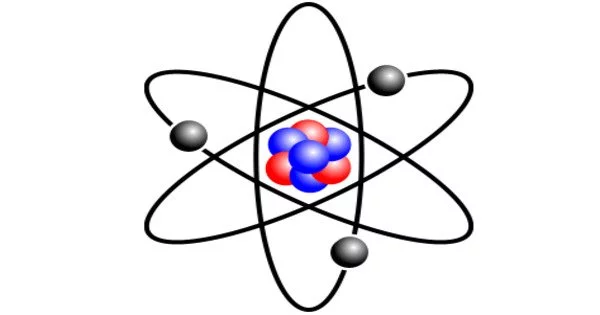
An atom of lithium is an atom of the chemical element lithium. Stable lithium is made up of three electrons held together by the strong force to a nucleus containing three protons and either three or four neutrons, depending on the isotope.
In its neutral state, the lithium atom is the chemical element lithium (Li). It is the third element in the periodic table and has an atomic number of 3, indicating that its nucleus contains three protons. In addition, the lithium atom has three electrons to balance the charge of the protons, resulting in a neutral atom.
A closed-form solution to the Schrödinger equation for the lithium atom has not been found, as it has not been found for the helium atom. However, approximations such as the Hartree-Fock method can be used to estimate the atom’s ground state energy and wavefunction. The quantum defect is a numerical value that represents the deviation from hydrogenic energy levels.
A lithium atom has the electronic configuration 1s2 2s1. This means it has two electrons in the first energy level (or shell) and one electron in the second. The innermost shell, known as the 1s shell, is closest to the nucleus and can only hold two electrons. The single electron of lithium is located in the outermost shell, the 2s shell.
Properties
- Atomic number: Lithium has an atomic number of 3, which means it has three protons in its nucleus.
- Atomic mass: The atomic mass of lithium is approximately 6.94 atomic mass units (amu). This value represents the average mass of lithium isotopes found in nature, taking into account their relative abundance.
- Electronic configuration: The electron configuration is 1s² 2s¹, indicating that it has two electrons in the first energy level (1s orbital) and one electron in the second energy level (2s orbital).
- Ionization energy: The first ionization energy of lithium is 5.39 electron volts (eV). This energy represents the amount of energy required to remove the most loosely bound electron from a neutral lithium atom, resulting in the formation of a Li+ ion.
- Melting and boiling points: Lithium has a relatively low melting point of 180.5 degrees Celsius (356.9 degrees Fahrenheit) and a boiling point of 1,342 degrees Celsius (2,448 degrees Fahrenheit). These temperatures reflect the phase changes of lithium from a solid to a liquid and from a liquid to a gas, respectively.
Lithium is an alkali metal that belongs to Periodic Group 1 (or 1A). Its low density, silvery-white appearance, and reactivity set it apart. Lithium is frequently found in compounds rather than as a pure element in nature. It is used in a variety of applications, including rechargeable batteries, ceramics, lubricants, and pharmaceuticals. Lithium has received a lot of attention in recent years because of its unique properties as a key component in lithium-ion batteries, which are widely used in electronic devices and electric vehicles.