An international scientific collaboration has made significant progress toward the realization of a nearly “green” zero-net-carbon technology that will efficiently convert carbon dioxide, a major greenhouse gas, and hydrogen into ethanol, which can be used as a fuel and has many other chemical applications. The study provides a “roadmap” for successfully navigating this difficult reaction and depicts the entire reaction sequence using theoretical modeling and experimental characterization.
The group, led by the US Department of Energy’s (DOE) Brookhaven National Laboratory, discovered that bringing cesium, copper, and zinc oxide together in close contact catalyzes a reaction pathway that converts carbon dioxide (CO2) into ethanol (C2H6O). They also figured out why this three-part interface works so well.
The research, which is described in a paper in the online edition of the Journal of the American Chemical Society and is featured on the cover of the journal, will spur further research into how to develop a practical industrial catalyst for selectively converting CO2 into ethanol. Such processes will result in technologies capable of recycling CO2 emitted by combustion and converting it into usable chemicals or fuels.
A group of researchers determined that bringing cesium, copper, and zinc oxide together into a close-contact configuration catalyzes a reaction pathway that transforms carbon dioxide into ethanol.
None of the three components studied in the study can catalyze the carbon dioxide (CO2)-to-ethanol (C2H6O). conversion alone or in pairs. However, when the trio is brought together in a specific configuration, the region where they meet opens a new route for the formation of carbon-carbon bonds, allowing the conversion of CO2 to ethanol. The well-tuned interplay between the cesium, copper, and zinc oxide sites is the key to this.
“Although much research has been done on the conversion of carbon dioxide to methanol, ethanol has many advantages over methanol. Ethanol is a more potent and safer fuel. However, due to the complexity of the reaction and the difficulty in controlling C-C bond formation, its synthesis is extremely difficult” Ping Liu, a chemist at Brookhaven National Laboratory, was the study’s corresponding researcher. “We now understand what kind of configuration is required to complete the transformation, as well as the roles that each component plays during the reaction. It’s a huge step forward.”
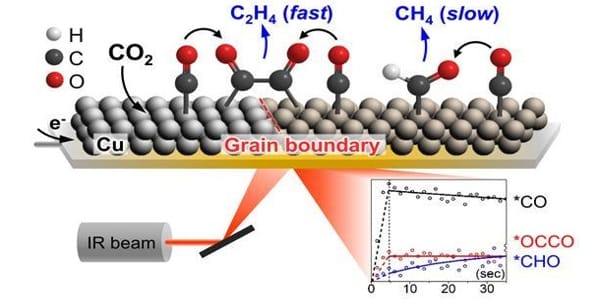
The interface is created by depositing trace amounts of copper and cesium onto a zinc oxide surface. To investigate the interfaces between the three materials, the researchers used an X-ray technique known as X-ray photoemission spectroscopy, which revealed a likely change in the reaction mechanism for CO2 hydrogenation when cesium was added. More information was obtained by employing two widely used theoretical approaches: “Density functional theory” calculations, a computational modeling method used to investigate the structures of materials, and “kinetic Monte Carlo simulation,” a computer simulation used to simulate reaction kinetics.
The group used the computing resources of Brookhaven National Laboratory’s Center for Functional Nanomaterials and Lawrence Berkeley National Laboratory’s National Energy Research Scientific Computing Center for this work, both of which are DOE Office of Science User Facilities.
One thing they discovered from the modeling is that cesium is an essential component of the active system. Ethanol cannot be produced without it. Furthermore, good coordination with copper and zinc oxide is essential. But there is still a lot to learn.
“There are many challenges to overcome before we can develop an industrial process that can convert carbon dioxide into usable ethanol,” said Brookhaven chemist José Rodriguez, who was involved in the research. “For example, there must be a clear way to improve ethanol production selectivity. Understanding the relationship between the nature of the catalyst and the reaction mechanism is a critical issue, and this study is at the forefront of that effort. We want to have a fundamental understanding of the process.”
Another goal of this research is to find an ideal catalyst for CO2 conversion to “higher” alcohols, which have two or more carbon atoms (ethanol has two) and are thus more useful and desirable for industrial applications and commodity goods production. The catalyst investigated in this study is advantageous because copper and zinc oxide-based catalysts are already widely used in the chemical industry for catalytic processes such as methanol synthesis from CO2.
The researchers intend to conduct additional research at Brookhaven’s National Synchrotron Light Source II, a DOE Office of Science User Facility that provides a unique suite of tools and techniques for the characterization of catalysts under operating conditions. There, they will delve deeper into the Cu-Cs-ZnO system and different-composition catalysts.