Flow batteries are a type of rechargeable electrochemical energy storage device that stores energy in external tanks of liquid electrolytes. They are well-known for their ability to provide large-scale, long-term energy storage solutions for renewable energy integration and grid stability. In a world-first experiment, a common food and medicine additive demonstrated that it can increase the capacity and longevity of a next-generation flow battery design.
According to a team of researchers from the Department of Energy’s Pacific Northwest National Laboratory, the flow battery, a design optimized for electrical grid energy storage, retained its capacity to store and release energy after more than a year of continuous charge and discharge.
The study, which was recently published in the journal Joule, describes the first use of a dissolved simple sugar called β-cyclodextrin, a starch derivative, to increase battery longevity and capacity. In a series of experiments, the scientists optimized the chemical ratio in the system until it produced 60% more peak power. They then repeatedly cycled the battery for more than a year, only stopping the experiment when the plastic tubing failed. The flow battery barely lost any of its activity to recharge during that time. This is the first laboratory-scale flow battery experiment to report continuous use for more than a year with minimal capacity loss.
This is a brand new approach to developing flow battery electrolyte. We demonstrated that a completely different type of catalyst can be used to accelerate energy conversion. Furthermore, because it is dissolved in the liquid electrolyte, there is no chance of a solid dislodging and fouling the system.
Wei Wang
In a process known as homogeneous catalysis, the -cyclodextrin additive is also the first to speed up the electrochemical reaction that stores and then releases the flow battery energy. This means that the sugar works while in solution rather than as a solid applied to a surface.
“This is a brand new approach to developing flow battery electrolyte,” said Wei Wang, a long-time PNNL battery researcher and the study’s principal investigator. “We demonstrated that a completely different type of catalyst can be used to accelerate energy conversion.” Furthermore, because it is dissolved in the liquid electrolyte, there is no chance of a solid dislodging and fouling the system.”

What is a flow battery?
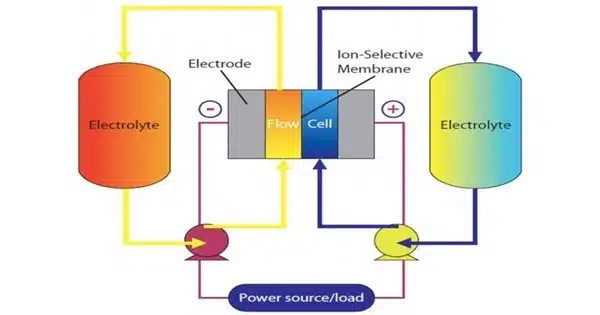
Flow batteries, as the name implies, are made up of two chambers, each filled with a different liquid. The batteries charge electrochemically and store energy in chemical bonds. When connected to an external circuit, they release energy that can be used to power electrical devices. Flow batteries are distinguished from solid-state batteries by the presence of two external supply tanks of liquid that are constantly circulating through them to supply the electrolyte, acting as the system’s “blood supply.” The more energy the flow battery can store, the larger the electrolyte supply tank.
If they are scaled up to the size of a football field or more, flow batteries can serve as backup generators for the electric grid. Flow batteries are one of the key pillars of a decarbonization strategy to store energy from renewable energy resources. Their advantage is that they can be built at any scale, from the lab-bench scale, as in the PNNL study, to the size of a city block.
Why do we need new kinds of flow batteries?
Large-scale energy storage acts as a kind of insurance policy against power grid disruption. When severe weather or high demand make it difficult to provide electricity to homes and businesses, energy stored in large-scale flow battery facilities can help minimize disruption or restore service. The demand for these flow battery facilities is only expected to increase as more electricity is generated using renewable energy sources such as wind, solar, and hydroelectric power. These intermittent power sources necessitate the storage of energy until it is required to meet consumer demand.
While there are numerous flow battery designs and commercial installations, existing commercial facilities rely on mined minerals such as vanadium, which is expensive and difficult to obtain. As a result, research teams are looking for effective alternative technologies that make use of more common materials that are easily synthesized, stable, and non-toxic.
“We cannot always dig the Earth for new materials,” said Imre Gyuk, director of energy storage research at the Department of Energy’s Office of Electricity. “Like the pharmaceutical and food industries, we need to develop a sustainable approach with chemicals that we can synthesize in large quantities.”
The work on flow batteries is part of a large program at PNNL to develop and test new technologies for grid-scale energy storage that will be accelerated with the opening of PNNL’s Grid Storage Launchpad in 2024.
A benign ‘sugar water’ sweetens the pot for an effective flow battery
The PNNL research team that developed this new battery design includes researchers with backgrounds in organic and chemical synthesis. These skills came in handy when the team chose to work with materials that had not been used for battery research, but which are already produced for other industrial uses.
“We were looking for a simple way to dissolve more fluorenol in our water-based electrolyte,” said first author Ruozhu Feng. “The -cyclodextrin contributed modestly to this, but its real benefit was this surprising catalytic ability.”
The researchers then collaborated with Yale University co-author Sharon Hammes-Schiffer, a leading authority on the chemical reaction underlying the catalytic boost, to explain how it works.
The sugar additive, as described in the research study, accepts positively charged protons, which helps balance out the movement of negative electrons as the battery discharges. The details are a little more complicated, but it’s as if the sugar sweetens the pot, allowing the other chemicals to finish their chemical dance.