Variable genomes of microbial bacteria living in a human mouth
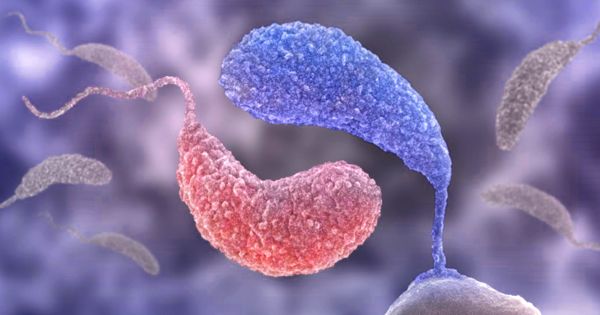
Bacteria are so small and small that it is difficult to classify the subgroups of bacteria that exist and the genes or metabolic abilities that cause them to survive in these ‘wrong’ sites. Researchers look more closely at the genomes of microbial species in the human mouth. Researchers looked at human oral microbiota and observed immense diversity in bacterial subpopulations residing in some parts of the mouth. In certain instances, the team was able to classify a handful of genes that could clarify the habitat specificity of a particular bacterial species.
Scientists from Harvard University studied the human oral microbiome and discovered impressive variability in bacterial subpopulations living in some mouth regions.
Bacteria also display very clear biogeography – certain bacteria are prevalent in certain places while others are absent – leading to big problems when applying microbiology to therapeutics or probiotics: how did the bacteria come to the wrong place? How can we apply the right bacteria to the right location when the biogeography is out of the way?
These problems, however, have one big obstacle: the bacteria are very small and multiple with very complex and complicated populations, causing major difficulties in understanding which subgroups of bacteria reside where and what genes or metabolic abilities cause them to survive in these ‘wrong’ sites.
Researchers led by Harvard University in a recent study published in Genome Biology looked at human oral microbiota and found impressive diversity in bacterial subpopulations living in some parts of the mouth.
“As microbial ecologists, we are fascinated by how bacteria can apparently divide any habitat into different niches, but as humans, we also have this innate curiosity about how microbes shape themselves within our bodies,” said lead author Daniel R. Utter, a Ph.D. candidate at the Department of Organism and Evolutionary Biology at Harvard University.
Latest advances in sequencing and bioinformatics have provided new avenues to uncover the structure of bacterial species. Utter and Colleen Cavanaugh, Edward C. Jeffrey Professor of Biology at the Department of Organism and Evolutionary Biology, Harvard University, has teamed up with researchers at the Marine Biological Laboratory, Woods Hole, University of Chicago, and the Forsyth Institute to apply these state-of-the-art sequencing and interpretation methods to better understand oral microbiota.
“The mouth is the perfect place to study microbial communities,” said co-author A. Murat Eren, Assistant Professor at the Department of Medicine at the University of Chicago. “It’s not only the beginning of the GI tract, but it’s also a very special and small environment that’s microbially diverse enough to really start answering interesting questions about microbiomes and their evolution.”
The mouth includes a surprising number of site-specific bacteria in various locations. For example, the microbes found on the tongue are somewhat different from the microbes found on the plaque on the teeth. “Your tongue bacteria are more like those that reside in someone else’s tongue than those that live in your throat or in your gums! “He said Eren.
The team scanned public databases and downloaded 100 genomes containing four species of bacteria usually found in the mouth, Haemophilus parainfluenzae and the three oral species of the Rothia gene, and used them as guides to examine their relatives sampled in hundreds of volunteer mouths of the Human Microbiome Project (HMP).
Using this newly established method, called metapangenomics, which combines pangenomes (the sum of all genes contained in a group of similar bacteria) with methagenomics (the study of complete DNA from all bacteria in the community), the researchers were able to carry out an in-depth study of the genomes of the microbes, which led to a surprising discovery.
“We’ve found an enormous amount of variability,” Utter said. “But we were shocked by the pattern of this variability across the different parts of the mouth; specifically, between the tongue, cheek, and tooth surfaces.”
For example, within a single microbe population, researchers found distinct genetic forms that were closely linked to a single, separate location within the mouth. In certain instances, the team was able to recognize a handful of genes that could clarify the unique habitat of a particular bacterial species. Applying metapangenomics, researchers were also able to distinguish unique ways in which free-living bacteria in people’s mouths varied from their lab-grown relatives.
“The resolution provided by these techniques—through a direct comparison of the genomes of ‘domesticated’ and ‘wild’ bacteria—allows us to dissect these gene differences by gene,” states Cavanaugh. “We have also been able to identify novel bacterial strains related to, but different from, those we have in culture.”
“Having identified some really strong bacterial candidates that could determine adaptation to a particular habitat, we would like to test these hypotheses experimentally,” said Cavanaugh. These results could theoretically be the secret to decoding selective probiotics, where scientists could use what has been discovered about the habitat needs of each microbe to engineer beneficial microbes to land in a designated habitat.
“The mouth is so easily accessible that people have been working on bacteria from the mouth for a long time,” said co-author Jessica Mark Welch, a research associate at the Marine Biological Laboratory.
“Every ecosystem we look at has very complicated, diverse bacterial populations, so why is that? “This is what Mark Welch said. “Understanding why these communities are so complex and how different bacteria interact will help us better understand how to fix a bacterial community that is damaging to our health, telling us which microbes need to be removed or added back in.”
This research and others like it will offer new insights into the role of oral microbes in human health. “The ability to identify specific genes behind habitat adaptation has been somewhat ‘holy grail’ in microbial ecology,” Utter said. “We are really excited about our contributions in this field. ”