Researchers have discovered that metabolic changes influence the formation of blood cells during embryonic development. They discovered a previously unknown metabolic switch that plays an important role in the development of different types of blood cells. This means that blood cell formation can be directed in the laboratory toward the production of natural killer cells, which will eventually be used in a new anti-cancer treatment.
Immunotherapy has heralded a thrilling new era in cancer treatment. The recent discovery and success of immune checkpoint blockade and chimeric antigen receptor (CAR) T cell adoptive cell transfer has sparked interest in using other immune cells, such as Natural Killer (NK) cells, to circumvent some of the limitations of CAR T cell therapy.
Researchers from Lund University in Sweden discovered that metabolic changes affect how blood cells form during embryonic development in a recent study. They discovered a previously unknown metabolic switch that plays an important role in the development of different types of blood cells. This means that blood cell formation can be directed in the laboratory toward the production of natural killer cells, which will eventually be used in a new anti-cancer treatment.
We discovered how to control the production of specific blood cells from iPS cells using metabolic factors. We intend to modify natural killer cells so that they can target and destroy specific cancers. We simply increased the availability of natural killer cells.
Niels-Bjarne Woods
In our bodies, approximately two billion natural killer cells circulate and play an important role in the body’s defense against cancer and infection. This has led researchers to believe that natural killer cells can be used in immunotherapy to treat the same diseases. As a result, we need to understand how these anti-cancer cells are generated from blood stem cells that originate in the embryo.
“We can reprogram cells from adult tissues, such as a hair follicle or a skin cell, to an embryonic-like state, resulting in iPS cells, or induced pluripotent stem cells. Because of their embryonic state, these cells have the potential to develop into a variety of cell types in the body, including blood cells “Niels-Bjarne Woods, Associate Professor at Lund University and the study’s corresponding author, agrees.
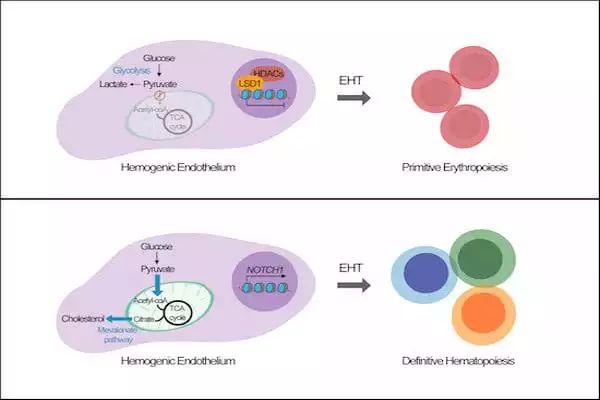
The research team discovered a metabolic switch in our system for generating blood cells from iPS cells that can activate the generation of specific blood cell types and, as a result, increase the production of natural killer cells.
“Our cells contain mitochondria, which act as power plants, delivering energy to the cells through chemical reactions. We could influence energy production and which types of blood cells were developed by feeding different substances to the cells. We conducted the experiments in the lab first, and then confirmed the findings in animals, achieving similar results” Niels-Bjarne Woods says.
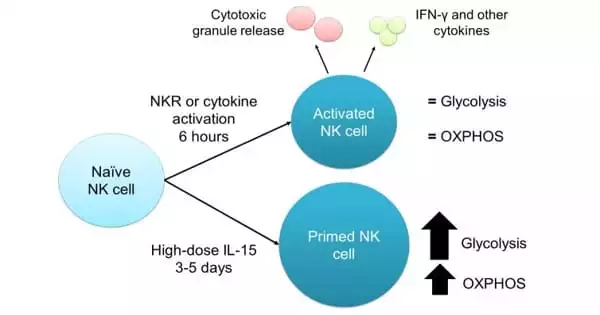
Increased mitochondrial activity resulted in significantly higher levels of natural killer cells. This demonstrates that metabolism plays an important role in blood development during the embryonic stage.
“We discovered how to control the production of specific blood cells from iPS cells using metabolic factors. We intend to modify natural killer cells so that they can target and destroy specific cancers. The next big hope for anti-cancer therapies is the transplantation of genetically modified natural killer cells derived from iPS cells” Niels-Bjarne Woods concludes. “We simply increased the availability of natural killer cells.”
The ability of the NK cell to recognize and rapidly kill tumor cells while causing no harm to healthy tissue suggests that it has the potential to be used in allogenic cancer immunotherapy. Indeed, several efforts have been made to develop clinical grade methods for generating large numbers of NK cells from a variety of sources, including peripheral blood, umbilical cord blood, induced pluripotent stem cells (iPSCs), and immortalized NK cell lines. These techniques allow for the manipulation of NK cells to maximize their antitumor potential, such as genetic engineering (CAR-NK) or NK cell pre-activation prior to adoptive transfer therapy.
Without prior sensitization, natural killer (NK) cells are the host’s first line of defense against tumors and viral infections. It is becoming increasingly clear that NK cells are members of the innate lymphoid cell (ILC) family. Other ILCs, which include ILC1s, ILC2s, ILC3s, and lymphoid tissue inducer (LTi) cells, are non-cytotoxic, tissue-resident cells that function to protect local microenvironments from tissue insults and to maintain homeostasis.