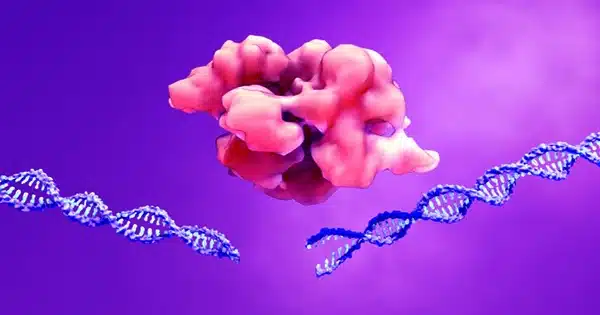
Tiny pores in the cell nucleus protect and preserve genetic material, which is essential for healthy aging. A team from the Max Planck Institute of Biophysics in Frankfurt am Main and the Synthetic Biophysics of Protein Disorder Group at Johannes Gutenberg University Mainz in Germany literally filled a hole in our understanding of the structure and function of these nuclear pores. The researchers discovered how inherently disordered proteins in the pore’s core may form a spaghetti-like movable barrier that allows critical cellular components through but inhibits viruses and other pathogens.
The nuclear membrane protects human cells’ genetic material inside the cell nucleus. The nucleus, as the cell’s control center, must be able to exchange vital messenger molecules, metabolites, or proteins with the remainder of the cell. As a result, around 2000 holes are created into the nuclear membrane, each of which contains approximately 1000 proteins.
For decades, researchers have been fascinated by the three-dimensional structure and function of these nuclear pores, which act as guardians of the genome, allowing substances required for cell control to pass while preventing pathogens or other DNA-damaging substances from entering. As a result, the nuclear pores may be compared to molecular bouncers, with each inspecting thousands of visitors every second. Only those who have an entrance ticket are allowed to pass.
How do the nuclear pores handle such a massive task? About 300 proteins attached to the pore scaffold protrude like tentacles deep into the central opening. Researchers had not known how these tentacles are structured or how they repel invaders until today. This is due to the fact that these proteins are disordered by nature and lack a clear three-dimensional structure. They are malleable and constantly moving, much like spaghetti in boiling water.
Microscopy and computer modeling combined
Because these intrinsically disordered proteins (IDPs) are constantly changing their structure, scientists are having difficulty deciphering their three-dimensional architecture and function. Most experimental approaches for imaging proteins only work with a specified 3D structure. Because the organization of the IDPs in the opening could not be determined, the central region of the nuclear pore has been represented as a hole thus far.
The team led by Gerhard Hummer, Director of the Max Planck Institute of Biophysics, and Edward Lemke, Professor for Synthetic Biophysics at Johannes Gutenberg University Mainz and Adjunct Director of the Institute of Molecular Biology Mainz, studied nuclear pore IDPs in living cells using a novel combination of synthetic biology, multidimensional fluorescence microscopy, and computer-based simulations.
“We used modern precision tools to mark several points of the spaghetti-like proteins with fluorescent dyes that we excite by light and visualize in the microscope,” adds Lemke. “Based on the glow patterns and duration, we were able to deduce how the proteins must be arranged.” “We then used molecular dynamics simulations to calculate how the IDPs are spatially organized in the pore, how they interact with one another, and how they move,” Hummer continues. We were able to see the gate to the control center of human cells for the first time.”
Dynamic protein network as transport barrier: Because they interact with each other and with the cargo, the tentacles in the transport pore exhibit completely different behavior than we previously observed. They travel indefinitely, much like the previously stated spaghetti in boiling water. So, instead of a hole, there is a shield of wiggly, spaghetti-like molecules in the center of the pore. Viruses and bacteria cannot pass through this filter. Other large cellular molecules required in the nucleus, on the other hand, can pass because they carry very specific signals. Pathogens, on the other hand, do not normally have an admission ticket. “By disentangling the pore filling, we enter a new phase in nuclear transport research,” says Martin Beck, a Max Planck Institute of Biophysics collaborator, and colleague.
“Knowing how the pores transport or block cargo will assist us in identifying errors.” After all, despite the barrier, some viruses manage to enter the cell nucleus,” Hummer concludes. “With our combination of methods, we can now study IDPs in greater depth to discover why, despite their error-prone nature, they are essential for certain cellular functions.” In reality, IDPs are ubiquitous in practically all species, but they pose the risk of forming aggregates throughout the aging process, which can contribute to neurological illnesses like Alzheimer’s,” Lemke explains. Researchers want to develop novel treatments or vaccinations that prevent viral infections and promote healthy aging by studying how IDPs work.