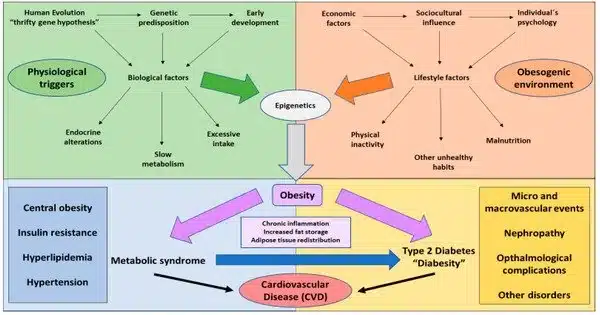
Certain genetic factors can contribute to a person’s proclivity for type 2 diabetes and obesity. These genetic factors may influence metabolic processes such as insulin production and sensitivity, energy expenditure, and fat storage. Certain gene variants can increase the likelihood of these conditions developing, but they do not ensure their occurrence. The rising prevalence of type 2 diabetes and obesity worldwide cannot be explained solely by genetic factors.
Researchers discovered an additive effect of genetic and socioeconomic risk on the prevalence of obesity and type 2 diabetes in analyses of more than 260,000 individuals’ genetic and area-level socioeconomic data.
According to new research led by researchers at Massachusetts General Hospital (MGH), a founding member of Mass General Brigham (MGB), socioeconomic and genetic factors likely interact in an additive manner to affect people’s risks of developing obesity and type 2 diabetes. The findings, which were published in Diabetes Care, suggest that interventions to reduce socioeconomic deprivation may reduce metabolic diseases at the individual and community levels, particularly among people who have a high genetic risk of metabolic disease.
Both genetic and socioeconomic factors have been shown to increase the risk of metabolic diseases, one intrinsic and unmodifiable and the other extrinsic and potentially modifiable, but the relative contributions of the two and the extent to which they may interact to impact a person’s risk are unknown.
We believe that this research calls for a whole-person approach to metabolic disease prevention and that public health interventions may be most impactful if targeted to those who also have elevated genetic risk.
Sara Cromer
To investigate, scientists looked at the independent and additive effects of genetic and socioeconomic risk in 26,737 and 223,843 European genetic ancestry participants from the Mass General Brigham Biobank and the UK Biobank, respectively, as well as 3,468 and 7,459 non-European ancestry participants from the respective biobanks. The researchers looked at people’s genetic data at millions of points across the genome, as well as information about their education, income, and employment in their area of residence. Because educational attainment had the strongest association with type 2 diabetes and obesity of all the area-level socioeconomic variables studied, it was chosen as the primary socioeconomic risk factor.
Results indicated that people in the highest quintile of both genetic and socioeconomic risk had a more than seven-fold higher prevalence of type 2 diabetes (22.2% vs. 3.1%) and a more than three-fold higher prevalence of obesity (69.0% vs. 20.9%) compared with those in the combined lowest risk quintiles.
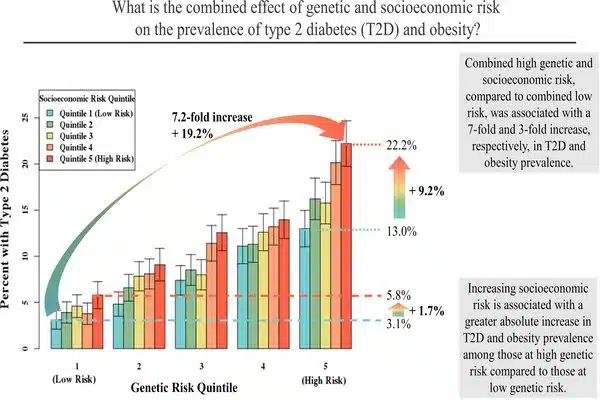
On an additive scale, there was a significant positive interaction between genetic and socioeconomic risk. This implies that the absolute increase in metabolic disease prevalence associated with unfavorable socioeconomic risk was much greater for those with higher genetic risk than for those with lower genetic risk. Negative area-level socioeconomic risk, for example, was associated with increased type 2 diabetes prevalence across the genetic risk spectrum, but the absolute increase in prevalence was greatest in those at highest genetic risk: +9.2% in the highest genetic risk quintile vs. +1.7% in the lowest genetic risk quintile. Overall, the combined effects of genetic and socioeconomic factors accounted for 13.2% and 16.7% of the prevalence of type 2 diabetes and obesity, respectively.
“We believe that this research calls for a whole-person approach to metabolic disease prevention and that public health interventions may be most impactful if targeted to those who also have elevated genetic risk,” says lead author Sara Cromer, MD, an Endocrinologist in the Department of Medicine at MGH and an Instructor at Harvard Medical School.
“The next steps in this research include expanding models to include more risk factors (such as lifestyle factors and behaviors), improving models for individuals of non-European ancestry, exploring the predictive value of area-level socioeconomic measures in diverse populations, and examining the gene-socioeconomic status interplay in regards to other outcomes.”
According to senior author Miriam Udler, MD, PhD, an endocrinologist in the department of Medicine at MGH, an investigator in the MGH Center for Genomic Medicine, and an assistant professor at Harvard Medical School, the study highlights not only the high prevalence of metabolic disease among individuals with both genetic and socioeconomic risk factors, but also that genetic risk for these diseases is not deterministic.
“People at high genetic risk who live in low-risk socioeconomic regions have the same rates of type 2 diabetes and obesity as people at low genetic risk who live in certain socioeconomic risk regions,” she says. “More research is needed to determine why this is the case.”