New research into catalyst design has revealed that hydrogen can be produced cleanly with much greater efficiency and at a lower cost than is currently possible with commercially available catalysts.
Oregon State University research into catalyst design has revealed that hydrogen can be produced cleanly with much greater efficiency and at a lower cost than is currently possible with commercially available catalysts. A catalyst is a substance that increases the rate of a chemical reaction without undergoing any permanent chemical change.
The findings are significant because hydrogen production is important for “many aspects of our lives, such as fuel cells for cars and the manufacture of many useful chemicals such as ammonia,” according to Zhenxing Feng, a chemical engineering professor at the Ohio State University College of Engineering who led the research. “It is also used in the refining of metals, the production of man-made materials such as plastics, and a variety of other applications.”
As part of their metabolic processes, certain photosynthetic microbes use light energy to produce hydrogen from water. Photobiological hydrogen production technology must overcome the inherent oxygen sensitivity of hydrogen-evolving enzyme systems because oxygen is produced alongside hydrogen. NREL researchers are addressing this issue by searching for naturally occurring organisms that are more oxygen tolerant and developing new genetic forms of organisms that can produce hydrogen in the presence of oxygen.
Catalysts are critical to promoting the water-splitting reaction by lowering the overpotential and, as a result, lowering the total cost of hydrogen production. Our first study in JACS Au laid the groundwork for us, and as demonstrated in our Science Advances article, we now have a better understanding of how to manipulate atoms on the surface to design catalysts with the desired structure and composition.
Zhenxing Feng
According to Feng, producing hydrogen by splitting water via an electrochemical catalytic process is cleaner and more sustainable than the traditional method of obtaining hydrogen from natural gas via a carbon dioxide-producing process known as methane-steam reforming. However, the higher cost of the greener technique has been a market barrier.
The new findings, which describe methods for designing catalysts that can significantly improve the efficiency of the clean hydrogen production process, were published in Science Advances and JACS Au.
Catalysts frequently undergo structural changes while facilitating reaction processes, according to Feng. Sometimes the changes are reversible, sometimes they are irreversible, and irreversible restructuring is thought to degrade a catalyst’s stability, resulting in a loss of catalytic activity and lowering reaction efficiency.

Feng, Maoyu Wang, an OSU Ph.D. student, and their colleagues studied the restructuring of catalysts in reaction and then manipulated their surface structure and composition at the atomic scale to achieve a highly efficient catalytic process for producing hydrogen.
An active phase of an amorphous iridium hydroxide-based catalyst demonstrated efficiency 150 times that of its original perovskite structure and nearly three orders of magnitude better than the common commercial catalyst, iridium oxide.
“We discovered at least two groups of materials that undergo irreversible changes and turned out to be significantly better hydrogen production catalysts,” Feng said. “This will allow us to produce hydrogen at a cost of $2 per kilogram, and eventually at a cost of $1 per kilogram. This is less expensive than current polluting processes and will contribute to the United States’ goal of zero emissions by 2030.”
Feng points out that the US Department of Energy’s Hydrogen and Fuel Cell Technologies Office has established benchmarks for technologies that can produce clean hydrogen for $2 per kilogram by 2025 and $1 per kilogram by 2030 as part of the Hydrogen Energy Earthshot target of reducing the cost of clean hydrogen by 80%, from $5 to $1 per kilogram, in a decade.
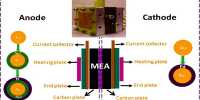
Feng’s group is working on water electrolysis technology for clean hydrogen production, which splits water using electricity generated from renewable sources. However, the efficiency of water splitting is low, he claims, owing to the high overpotential (the difference between an electrochemical reaction’s actual and theoretical potential) of one key half-reaction in the process, the oxygen evolution reaction, or OER.
“Catalysts are critical to promoting the water-splitting reaction by lowering the overpotential and, as a result, lowering the total cost of hydrogen production,” Feng explained. “Our first study in JACS Au laid the groundwork for us, and as demonstrated in our Science Advances article, we now have a better understanding of how to manipulate atoms on the surface to design catalysts with the desired structure and composition.”
The National Science Foundation-funded Feng’s research through the Northwest Nanotechnology Infrastructure site at Oregon State University, and the Department of Energy also contributed.