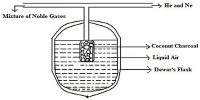
Difference between Main energy shell and Sub-Shell
Main energy shell
i. Is a group of atomic orbitals with the same value of principal quantum number n.
ii. The number of electrons that can reside in a shell is equal to 2n2.
iii. Main energy shell are identified by the letters K, L, M, N etc corresponding to principle quantum numbers (n) 1, 2, 3, 4 etc.
Sub-Shell
i. Electron shell are made up of one or more electron sub-shells or sublevels, which have two or more orbital with the same angular momentum quantum number l.
ii. Each sub-shell can hold 2 (2l + I) electrons.
iii. Electron sub-shells are identified by the letter s, p, d, f, g, h etc. corresponding to the azimuthal quantum numbers (l – values) 0, 1, 2, 3, 4, 5 etc.