The volume, V litres, of one mole of CO2 gas was measured as the pressure of p atmospheres (atm) was varied at a constant temperature of 313 K.
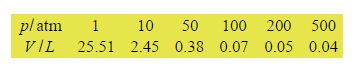
Manually plot p versus V and also plot log(p) against log( V). Comment upon which of the plots is most useful for displaying the data.
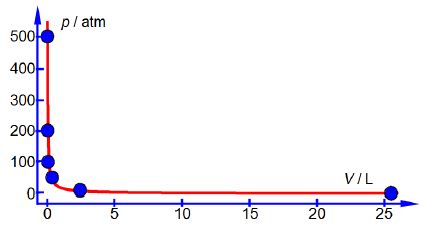
Pressure against volume plot for CO2
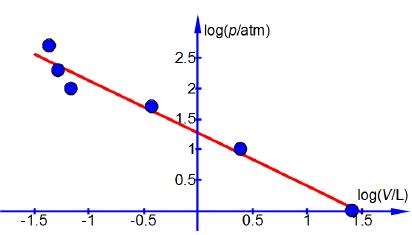
Log (p) versus log (v) plot for CO2
The data covers a wide range of values and clearly the log-log plot allows the data to be viewed more clearly and it also would allow any “rogue” points in the pressure volume experimental data to be noted for remeasuring.