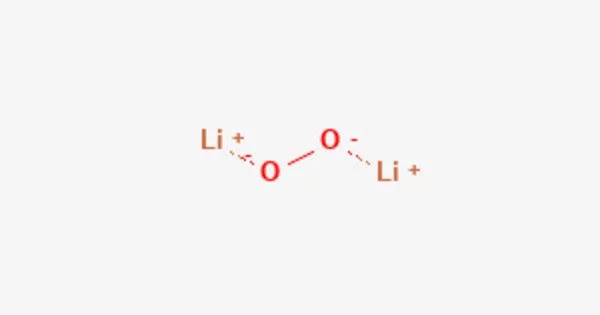
Lithium peroxide is a white, crystalline compound with the chemical formula Li2O2. It is a strong oxidizing agent and has been used as an oxygen source in breathing masks, spacesuits, and other applications requiring a source of oxygen. The compound is relatively unstable and can be dangerous in certain conditions, such as when it comes into contact with organic materials.
Properties
Lithium peroxide is an inorganic compound. It is a white, nonhygroscopic solid. Because of its high oxygen: mass and oxygen: volume ratios, the solid has been used to remove CO2 from the atmosphere in spacecraft.
- Chemical formula: Li2O2
- Molar mass: 45.881 g/mol
- Appearance: fine, white powder
- Odor: odorless
- Density: 2.31 g/cm3
- Melting point: Decomposes to Li2O at ~340°C
- Boiling point: NA
- Solubility in water: soluble[vague]
- Crystal structure: hexagonal

Preparation
It is prepared by the reaction of hydrogen peroxide and lithium hydroxide. This reaction initially produces lithium hydroperoxide:
LiOH + H2O2 → LiOOH + H2O
This lithium hydroperoxide has also been described as lithium peroxide monoperoxohydrate trihydrate (Li2O2·H2O2·3H2O). Dehydration of this material gives the anhydrous peroxide salt:
2 LiOOH → Li2O2 + H2O2
Li2O2 decomposes at about 450 °C to give lithium oxide:
2 Li2O2 → 2 Li2O + O2
The structure of solid Li2O2 has been determined by X-ray crystallography and density functional theory. The solid features an eclipsed “ethane-like” Li6O2 subunits with an O-O distance of around 1.5 Å.
Uses
It is used as an oxygen source in life support systems and as a component of oxygen candles, which are used to provide emergency oxygen in submarines, aircraft, and other closed environments. It is also being studied as a cathode material for lithium-air batteries.
It is used in air purifiers where weight is important, e.g., spacecraft to absorb carbon dioxide and release oxygen in the reaction:
Li2O2 + CO2 → Li2CO3 + 1⁄2 O2
It absorbs more CO2 than the same weight of lithium hydroxide and has the added benefit of releasing oxygen. Furthermore, unlike most other alkali metal peroxides, it is not hygroscopic. The reversible lithium peroxide reaction serves as the foundation for a prototype lithium-air battery. Using oxygen from the atmosphere allows the battery to avoid storing oxygen for its reaction, reducing battery weight and size.
The successful combination of a lithium-air battery overlain with an air-permeable mesh solar cell was announced by The Ohio State University in 2014. The combination of two functions in one device (a “solar battery”) is expected to significantly reduce costs when compared to separate devices and controllers as is currently used.