Mitochondrial function, specifically its role in cellular redox/oxidative balance, is critical in controlling cell life and death and has been linked to a variety of human pathologies. Mitochondria play a critical role in the regulation of cell death during oxidant accumulation. A recent discovery provides researchers with a new avenue to investigate oxidative stress and its detrimental effects in aging, cancer, and other diseases.
Mitochondria play an important role in the production of reactive oxygen species, which are now thought to be the primary pathogenic agent in many diseases. A vicious circle of oxidative stress and cellular structure damage can result in either cell death via apoptosis or cellular energetic decline and aging.
Early mitochondrial involvement in apoptosis includes the expression of pro-apoptotic factors, the release of cytochrome c from the inter-membrane space, and the opening of the permeability transition pore: cytochrome c release appears to precede pore opening. Somatic mutations (deletions) of mitochondrial DNA caused by oxygen radicals are thought to be the primary cause of energy decline in the mitochondrial theory of ageing; experimentally, Complex I appears to be the most affected.
We identify a key molecule that transports glutathione, the body’s main antioxidant, into the cell’s mitochondria, where free radicals are produced in large quantities.
Kivanç Birsoy
Many of the processes that keep us alive endanger us. Free radicals are unstable molecules that steal electrons from other molecules produced by energy-producing chemical reactions in our cells, for example. When free radicals are produced in abundance, they can cause collateral damage, potentially triggering malfunctions such as cancer, neurodegeneration, or cardiovascular disease.
Cells address this issue by producing antioxidants, which are compounds that neutralize free radicals. In a new study, Rockefeller University researchers identify a key molecule that transports glutathione, the body’s main antioxidant, into the cell’s mitochondria, where free radicals are produced in large quantities. The discovery, which was published in Nature, opens up new avenues for research into oxidative stress and its harmful effects.
“Now that we’ve identified a potential transporter, we can control the amount of glutathione that enters mitochondria and study oxidative stress at its source,” says Kivanç Birsoy, Chapman Perelman Assistant Professor at The Rockefeller University.
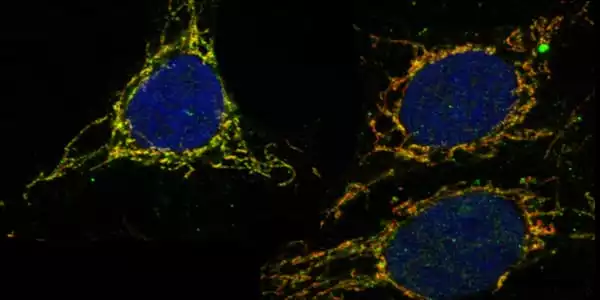
The shuttle into the mitochondria
To avoid oxidative stress, cells must maintain a proper balance of free radicals and antioxidants within their mitochondria, which is where energy is produced. Because glutathione is produced outside of mitochondria, in the cell’s cytosol, the researchers were curious as to how it gets into these tiny powerhouses in the first place.
Birsoy’s team studied protein expression in cells in response to glutathione levels in order to shed light on this process. Birsoy explains, “We hypothesized that glutathione is shuttled by a transporter protein whose production is regulated by glutathione.” “If we reduce glutathione levels, the cell should compensate by upregulating the transporter protein.”
The analysis pointed to SLC25A39, a protein in the mitochondrial membrane with an unknown function. The researchers discovered that inhibiting SLC25A39 reduced glutathione levels within the mitochondrion while having no effect on glutathione levels elsewhere in the cell. Other studies have shown that mice cannot survive without SLC25A39. Red blood cells in animals engineered to lack this protein die quickly from oxidative stress due to their inability to bring glutathione into mitochondria.
The discovery of the transporter could lead to a better understanding of a variety of oxidative stress-related disease pathways, including those involved in aging and neurodegeneration. “These conditions may be treated or prevented if antioxidant transport into mitochondria is stimulated,” Birsoy says.
Furthermore, the researchers are investigating whether SLC25A39 could be used as a cancer drug target by assisting in the induction of lethal oxidative stress in tumor cells. “In cancer, we’d want to prevent antioxidants from entering mitochondria, and the transporter protein could be our way of doing that,” Birsoy says.