According to new research, devices made of readily available oxide and carbon-based materials can produce clean hydrogen from water over several weeks. The findings, led by Dr. Virgil Andrei, a Research Fellow at St John’s College, University of Cambridge, and academics from Imperial College London, could help solve one of the key issues in solar fuel production, where current earth-abundant light-absorbing materials are limited in either performance or stability.
Underexplored materials for light harvesting
Hydrogen fuel will be critical in the transition to full decarbonisation and the achievement of the UK’s goal of net-zero emissions by 2050. With the majority of hydrogen currently supplied by fossil fuels, researchers are now working to develop more sustainable hydrogen generation methods. Making devices that can harvest sunlight and split water to produce green hydrogen is one way to accomplish this.
While many light-absorbing materials have been tested for green hydrogen production, the majority of them degrade rapidly when submerged in water. Perovskites, for example, are the most rapidly growing materials in terms of light-harvesting efficiency, but they are unstable in water and contain lead. As a result of the risk of leakage, researchers have been working to develop lead-free alternatives.
Bismuth oxyiodide (BiOI) is a non-toxic semiconductor alternative that has been overlooked for solar fuel applications due to its poor water stability. However, based on previous findings into the potential of BiOI, researchers decided to revisit the material’s promise for the production of green hydrogen.
Bismuth oxyiodide is an intriguing photoactive material with energy levels in the proper positions for water splitting. We demonstrated a few years ago that BiOI solar cells are more stable than those that use cutting-edge perovskite light absorbers. We wanted to see if we could apply that stability to green hydrogen production.
Dr. Robert Hoye
Dr. Robert Hoye, Lecturer in the Department of Materials at Imperial College London, explained: “Bismuth oxyiodide is an intriguing photoactive material with energy levels in the proper positions for water splitting. We demonstrated a few years ago that BiOI solar cells are more stable than those that use cutting-edge perovskite light absorbers. We wanted to see if we could apply that stability to green hydrogen production.”
Professor Judith Driscoll, Department of Materials Science and Metallurgy, University of Cambridge, said: “We have been working on this material for some time, due to its wide-ranging potential applications, as well as its simplicity of fabrication, low toxicity and good stability. It was great to combine the expertise of the different research groups across Cambridge and with Imperial.”
Breakthrough in solar fuel production
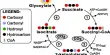
The researchers developed devices that mimicked the natural photosynthesis process that occurs in plant leaves, but instead of sugars, they produce fuels such as hydrogen. These artificial leaf devices, made of BiOI and other environmentally friendly materials, harvest sunlight to produce O2, H2, and CO.
By sandwiching BiOI between two oxide layers, researchers were able to improve the stability of these artificial leaf devices. The strong oxide-based device structure was then coated with a water-repellent graphite paste to keep moisture out. This increased the stability of the bismuth oxyiodide light-absorbing pixels from a few minutes to several months, including the time the devices were stored.
This is a significant finding that transforms BiOI into a viable light harvester for stable green hydrogen production.

“These oxide layers improve the ability to produce hydrogen compared to stand-alone BiOI,” said Dr Robert Jagt (Department of Materials Science and Metallurgy, University of Cambridge), one of the co-lead authors.
Researchers discovered that artificial leaf devices with multiple light harvesting areas (called ‘pixels’) performed better than conventional devices with a single larger pixel of the same total size. This discovery could make it much easier and faster to scale up novel light harvesters for sustainable fuel production.
Dr. Virgil Andrei, a co-lead author from Cambridge’s Department of Chemistry, explains: “Even if some pixels are defective, we were able to disconnect them so that they do not affect the rest of the pixels. As a result, we were able to maintain the performance of the small pixels over a larger area.” Because of this improved performance, the device was able to not only produce hydrogen but also reduce CO2 to synthesis gas, an important intermediate in the industrial synthesis of chemicals and pharmaceuticals.
Looking to the future
The results show that these new devices have the potential to outperform existing light absorbers. The new techniques for making BiOI artificial leaf devices more stable can now be applied to other novel systems, assisting in their commercialization.
“This is an exciting development! At the moment, few solar fuel systems demonstrate stability suitable for real-world applications. With this work, we take another step toward establishing a circular fuel economy” Prof Erwin Reisner (Department of Chemistry, Cambridge) is one of the corresponding authors.