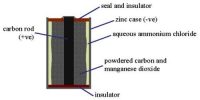
A thermo-chemical equation is a chemical equation for a reaction in which the enthalpy of reaction for the molar amounts stated is written directly after the equation.
In a thermo-chemical equation, it is important to note state symbols (solid, liquid or gas) because the enthalpy change, ΔH, depends on the phase of the substances.
Manipulating Thermo-chemical Equations: The following are two important rules for manipulating thermo-chemical equations:
i. When a chemical equation is reversed, the value of ΔH is reversed in sign. For example,
Sr(s) + ½ O2 (g) → SrO(s) ΔH = -592 kJmol-1
SrO(s) → Sr(s) + ½ O2 (g) ΔH = +592 kJmol-1
ii. When a thermo-chemical equation is multiplied by any factor, the value of AH for the new equation is obtained by multiplying the AH in the original equation by that same factor. For example,
Sr(s) + ½ O2 (g) → SrO(s) ΔH = -592 kJmol-1
2 Sr(s) + O2 (g) → 2 SrO(s) →H = -1184 kJmo1-1