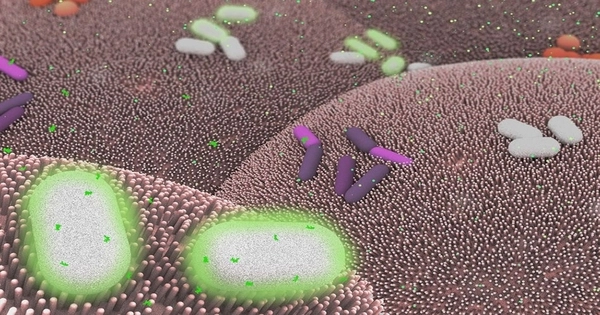
The small intestine’s primary function is to digest, absorb, and propel food along its length. Most clinically significant small intestine infections will disrupt these functions. Diarrhea is a common symptom, but there may also be bleeding, bloating, nausea, vomiting, abdominal pain, and even signs of complete abdominal obstruction.
According to Weill Cornell Medicine researchers, a common gut bacterium that can spread throughout the body and cause a serious infection resists natural immune defenses and antibiotics by enhancing its protective outer layer, known as the cell envelope. The discovery suggests new ways to combat these bacterial infections.
The study, published in mBio, sheds light on some of the underlying changes that may occur when Enterococcus faecalis (E. faecalis) populations move through the epithelial cells lining the intestine and escape to other body sites.
“Systemic infections with E. faecalis can be lethal because this microbe has a remarkable ability to adapt to different environments and resist treatments,” said principal investigator Dr. Diana K. Morales, assistant professor of microbiology and immunology in obstetrics and gynecology at Weill Cornell Medicine. Those who are taking antibiotics or have compromised immune systems, which allow E. faecalis overgrowth in the intestine, are at risk of developing these infections. Understanding how E. faecalis moves out of the gut and spreads may one day help scientists find small molecules to stop the bacterium’s extra-intestinal dissemination, preventing dangerous infections.
We are interested in identifying pharmacological approaches that can target these specific pathways with the goal of one day helping patients better fight infections caused by this gut microbe.
Dr. Morales
The mechanism by which the bacterium can move from the intestine to other organs is largely unknown. According to Dr. Morales, researchers have discovered two distinct populations of the same bacterium species. One population develops traits that allow it to pass through the intestinal barrier, acquiring advantageous antimicrobial resistance, while the other population remains stationary.
Previously, the researchers discovered that motile E. faecalis produces polysaccharides, which are molecules formed by sugar chains and allow the bacterium to aggregate or clump together. “These bacteria appear to develop the ability to move when they aggregate,” Dr. Morales said.
In the current study, the investigators, including lead author Dr. Yusibeska Ramos, a research associate in obstetrics and gynecology, found that the motile form of E. faecalis has a cell envelope containing increased amounts of glycolipids, which are fat molecules linked with a carbohydrate. Enhanced production of cell envelope glycolipids appears to help the bacterium to resist extracellular stressors. These stressors include the antimicrobial agent daptomycin, a common treatment for E. faecalis infection, and ?-defensins, small molecules intestinal epithelial cells produce to deter infection.
The researchers also discovered that genetic mutations that inhibit glycolipid production made E. faecalis more vulnerable to these stressors and decreased the bacterium’s ability to penetrate cell surfaces and move through intestinal epithelial cells.
The researchers’ next step will be to test additional in vivo models to see if the molecular pathways discovered in this study are required for the bacterium to exit the intestine. “We are also interested in identifying pharmacological approaches that can target these specific pathways with the goal of one day helping patients better fight infections caused by this gut microbe,” said Dr. Morales.