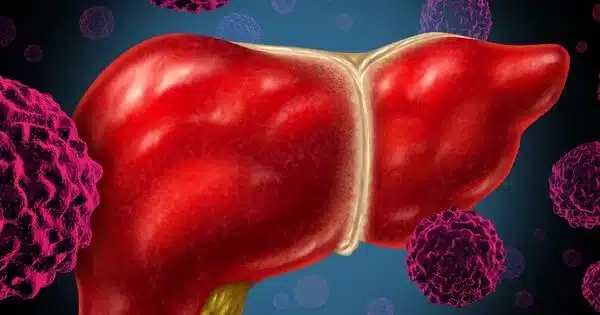
The development of novel artificial intelligence-based blood tests for the early detection of liver cancer would be a significant step forward in medical diagnostics. Early detection is critical for improving cancer patients’ prognoses and treatment outcomes. AI algorithms have demonstrated significant promise in analyzing complex data patterns and identifying disease markers in a variety of medical fields. Researchers may be able to discover specific biomarkers or patterns in the blood that indicate the presence of liver cancer in its early stages by applying AI techniques to blood tests.
In a new study of 724 people, a novel artificial intelligence blood testing technology developed and used by Johns Hopkins Kimmel Cancer Centre researchers to successfully detect lung cancer in a 2021 study has now detected more than 80% of liver cancers.
DELFI (DNA evaluation of fragments for early interception) is a blood test that detects fragmentation changes in DNA from cancer cells shed into the bloodstream, also known as cell-free DNA (cfDNA). In the most recent study, researchers used DELFI technology to detect hepatocellular cancer (HCC), a type of liver cancer, in blood plasma samples collected from 724 people in the United States, the European Union (E.U.), and Hong Kong.
According to the researchers, this is the first genome-wide fragmentation analysis that has been independently validated in two high-risk populations and across different racial and ethnic groups with different causes of liver cancer. Their findings were published in Cancer Discovery on November 18 and presented at the American Association for Cancer Research Special Conference: Precision Prevention, Early Detection, and Interception of Cancer on November 19.
Increased early detection of liver cancer could save lives, but currently, available screening tests are underutilized and miss many cancers.
Victor Velculescu
It is estimated that 400 million people worldwide are at higher risk of developing HCC because of cirrhosis from chronic liver diseases including chronic viral hepatitis or non-alcoholic fatty liver disease, according to a worldwide analysis of the burden of liver disease (J. Hepatology, 2019).
“Increased early detection of liver cancer could save lives, but currently available screening tests are underutilized and miss many cancers,” says Victor Velculescu, M.D., Ph.D., professor of oncology and co-director of the Cancer Genetics and Epigenetics Program at the Johns Hopkins Kimmel Cancer Center, who co-led the study with Zachariah Foda, M.D., Ph.D., gastroenterology fellow, Akshaya Annapragada, M.D./Ph.D. student, and Amy Kim, M.D., assistant professor of medicine at the Johns Hopkins University School of Medicine.
According to Foda, of the 724 plasma samples studied, 501 were collected in the United States and the European Union and included samples from 75 people with HCC to train and validate the machine learning model, a type of artificial intelligence that uses data and algorithms to improve accuracy. An additional 223 plasma samples from Hong Kong residents were analyzed for validation, including samples from 90 people with HCC, 66 with hepatitis B virus (HBV), 35 with HBV-related liver cirrhosis, and 32 with no underlying risk factors.
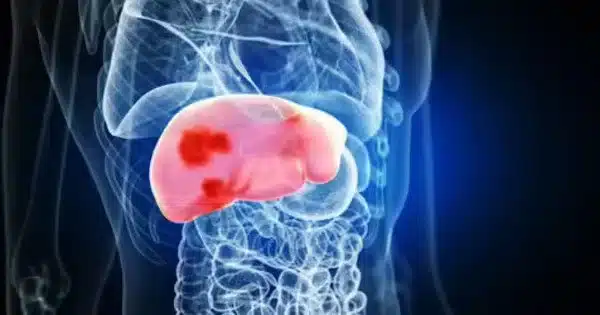
The DELFI technology employs a blood test to assess how DNA is packaged inside the nucleus of a cell by examining the size and amount of cell-free DNA present in the circulation from various regions of the genome. Healthy cells pack DNA like a well-organized suitcase, with different regions of the genome carefully placed in different compartments. Cancer cell nuclei, on the other hand, are more disorganised suitcases, with items from across the genome thrown in haphazardly. When cancer cells die, they release DNA fragments into the bloodstream in a chaotic manner.
DELFI detects cancer by examining millions of cfDNA fragments for abnormal patterns, such as the size and amount of DNA in various genomic regions. According to the researchers, the DELFI approach only requires low-coverage sequencing, making this technology cost-effective in a screening setting.
The latest study used cfDNA fragments isolated from plasma samples to perform the test, which has previously been shown to accurately classify lung cancer. They created a DELFI score by analyzing fragmentation patterns across each sample.
Scores were low for cancer-free people with viral hepatitis or cirrhosis (median DELFI scores were 0.078 and 0.080, respectively), but 5 to 10 times higher for the 75 HCC patients in the US/EU samples, with high scores observed across all cancer stages, including early-stage disease (DELFI scores for Stage 0 = 0.46, Stage A = 0.61, Stage B = 0.83, and Stage C = 0.92). Furthermore, the test detected changes in fragmentation in the content and packaging of liver cancer genomes, including from genome regions associated with liver-specific activity.
The DELFI technology detected liver cancers at their earliest stages, with an overall sensitivity — or ability to accurately detect a cancer — of 88% and a specificity of 98%, which means it almost never incorrectly provided a false positive result, among people at average risk. In samples collected from those at high risk of HCC, the test had 85% sensitivity and 80% specificity.
“Currently, due to accessibility and suboptimal test performance, less than 20% of the high-risk population is screened for liver cancer. When compared to the standard blood test available, this new blood test can double the number of liver cancer cases detected and increase early cancer detection,” says Kim, co-senior author on the study.
According to the researchers, the next step is to validate this approach in larger studies for clinical use. According to the American Cancer Society, more than 800,000 people are diagnosed with liver cancer each year, and it is the leading cause of cancer deaths worldwide.