Splitting water into hydrogen and oxygen is an alternative to fossil fuels, but filtered water is a vital option. Solar, wind, and sea power can soon combine to create clean-burning hydrogen fuel, according to a team of Penn State researchers. The team has incorporated water purification technologies into a recent test-of-concept design for a seawater electrolyzer that uses electrical current to separate hydrogen and oxygen into water molecules.
According to Bruce Logan, Professor of Environmental Engineering at Kappe and Professor of Environmental Engineering at Evan Pugh University, this modern process of “seawater splitting” could make it easier to convert wind and solar energy into a storable and portable power.
Researchers have integrated water purification technology into a new proof-of-concept design for a seawater electrolyzer that uses an electric current to split apart the hydrogen and oxygen in water molecules.
“Hydrogen is a great fuel, but you have to make it,” Logan said. “The only sustainable way to do that is to use renewable energy and produce it from water. You also need to use water that people do not want to use for other things, and that would be seawater. So, the holy grail of producing hydrogen would be to combine the seawater and the wind and solar energy found in coastal and offshore environments.”
Despite the availability of seawater, it is not widely used to separate water. Until the water is desalinated before hitting the electrolyzer—an costly extra step—the chloride ions in seawater turn into poisonous chlorine gas that degrades the machinery and sows into the setting.

Scientists Le Shi and Bruce Logan examine their newly designed seawater electrolyzer
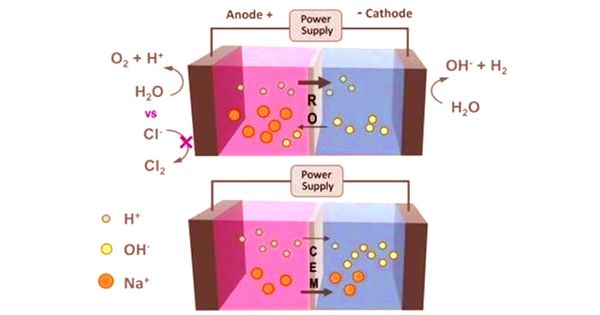
To avoid this, researchers have inserted a thin, semipermeable membrane, originally designed for the purification of water in the reverse osmosis (RO) treatment process. The RO membrane replaced the ion-exchange membrane typically used in electrolysers. “The idea behind RO is that you put a really high pressure on the water and push it through the membrane and keep the chloride ions behind,” Logan said.
In an electrolyzer, the seawater would no longer be pumped into the RO membrane but would be contained in it. The membrane is used to help distinguish reactions that occur near two submerged electrodes-a positively charged anode and a negatively charged cathode-connected by an external power source. When the electricity is switched on, the water molecules tend to divide at the anode, emit tiny hydrogen ions called protons, and produce oxygen gas. The protons then travel through the membrane and merge to form hydrogen gas with the electrons at the cathode.
With the RO membrane inserted, seawater is held on the cathode side, and the chloride ions are too high to travel through the membrane to enter the anode, stopping the development of chlorine gas. But in the water-splitting process, Logan observed, other salts are deliberately dissolved in the water to help make it conductive. The ion-exchange membrane, which channels ions by an electrical charge, helps salt ions to pass through. The RO membrane is not.
“RO membranes inhibit salt motion, but the only way you generate current in a circuit is because charged ions in the water move between two electrodes,” Logan said. For the passage of larger ions limited by the RO membrane, researchers wanted to see if there were sufficiently tiny protons flowing through the pores to hold the electrical current strong.
“Basically, we had to show that what looked like a dirt road could be an interstate,” Logan said. “We had to prove that we could get a high amount of current through two electrodes when there was a membrane between them that would not allow salt ions to move back and forth.”
Through a series of experiments recently published in Energy & Environmental Science, researchers tested two commercially available RO membranes and two cation-exchange membranes, a type of ion-exchange membrane that enables all positively charged ions to pass inside the structure. Each was evaluated for membrane resistance to ion movements, the amount of energy required to complete reactions, hydrogen and oxygen gas generation, chloride ion interaction, and membrane deterioration.
Logan explained that while one RO membrane turned out to be a “dirt road,” the other was doing well in contrast to the cation-exchange membranes. Researchers are now researching why there has been such a disparity between the two RO membranes. “The idea can work,” he said. “We do not know exactly why these two membranes have been functioning so differently, but that is something we are going to figure out.”
Recently, researchers have obtained a $300,000 grant from the National Science Foundation (NSF) to continue studies into marine water electrolysis. Logan expects that their study will play a vital role in reducing carbon dioxide emissions around the world.
“The world is looking for renewable hydrogen,” he said. “For example, Saudi Arabia has planned to build a $5 billion hydrogen facility that is going to use seawater. Right now, they have to desalinate the water. Maybe they can use this method instead.”