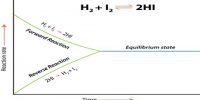
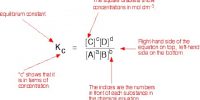
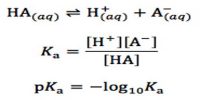Haber’s Process
Ammonia is mainly used as a source of nitrogen fertilizer, in nitric acid production and in nitrogen containing pharmaceuticals. Ammonia is commercially produced in industries from the gaseous elements nitrogen and hydrogen in air by means of Haber’s process. Ammonia formation reaction is an equilibrium reaction.
N2 (g) + 3H2 (g) ↔Fe↔ 2NH3 (g) [ΔH0f = –22.0 kcal/mole]
The forward reaction is accompanied by decrease in the number of moles of reactants and according to Le Chatlier’s principle, an increase in pressure favors such a reaction and shifts the equilibrium towards the product formation direction.
Therefore, nearly 300-500 atm pressure is applied on 3:1 mole ratio of H2 : N2 gas mixture in the reaction chamber for maximum yield of ammonia. The ammonia formation reaction is exothermic. By Le Chatlier’s principle, increase in temperature favours decomposition reaction of ammonia. However, at low temperature the time to reach the equilibrium becomes very long. Hence an optimum temperature close to 500°C-550°C is maintained. Iron catalyst is chosen to speed up the attainment of the equilibrium concentration of ammonia. In order to maintain the equilibrium conditions, steam is passed to remove away the ammonia as and when it is formed so that the equilibrium remains shifted towards the product side. The maximum yield of ammonia is nearly 37%.