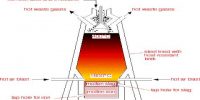
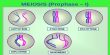
Hybridization is the concept of intermixing of the orbitals of an atom having nearly the same energy to give exactly equivalent orbitals with the same energy, identical shapes and symmetrical orientations in space.
The new equivalent orbitals formed are known as the hybrid orbitals or hybridized orbitals. Hybrid orbitals have properties entirely different from the properties of the original orbitals from which they have been obtained.
Features regarding Hybridization
(i) Orbitals involved in hybridization should have nearly the same energy.
(ii) The orbitals of one and the same atom participate in hybridization.
(iii) The number of hybrid orbitals formed is equal to the number of hybridizing orbitals.
(iv) The hybrid orbitals are all equivalent in shape and energy.
(v) A hybrid orbital which is taking part in bond formation must contain one electron in it.
(vi) Due to the electronic repulsions between the hybrid orbitals, they tend to remain at the maximum distance apart.
(vii) The head on overlap of atomic orbitals give sigma (σ) bonds.
(viii) The sidewise or lateral overlap of atomic orbitals give pi (π) bonds.