Regnault’s method
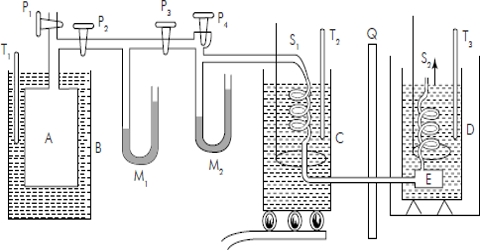
The method is based on the measurement of the mass of a known volume of gas under the definite condition of temperature and pressure. It is a technique for measuring gas density by evacuating and weighing a glass bulb of known volume, admitting gas at a known pressure, and reweighing. Two glass bulbs, each of 2 L capacity, are first dried, evacuated, and suspended from the two arms of a balance (Figure: Regnault’s apparatus for determination of density of a gas).
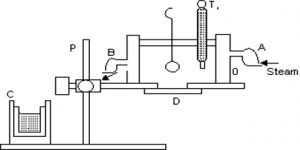
Principle: In this method vapor densities of gases are determined by direct weighing. The vapor density of a gas is the ratio of the mass of a certain volume of a gas to the mass of the same volume of hydrogen at the same temperature & pressure.
The bulbs are counterpoised in the balance. One of the bulbs says A, is then filled with gas under known pressure and again counterpoised against bulb B. adding necessary weights on the pan from which B is suspended.
Procedure: In this method, two hollow glass globes of the same capacity, same mass, and the same size are taken. They are evacuated and suspended by two sides of a physical balance. Now one of the glass globes is filled with a gas whose vapor density is to be found. Then the mass of the globe is measured. The difference in the filled globe and empty globe gives the mass of the gas around the globe. Now the globe is evacuated again and filled with hydrogen gas at the same temperature and pressure.
This additional weight necessary for balancing the arms is the mass of gas taken in bulb A. The temperature of the surroundings is kept uniform. The volume of bulb A is obtained from the mass of water necessary to till the bulb and the density of water at the temperature of the experiment. The determination must be carried out at a constant known temperature and the result corrected to standard temperature and pressure.
From the mass and volume of the gas, the density may be calculated, and knowing the pressure and temperature the molecular mass of the gas is obtained. The bulb B is used as a counterpoise in order to minimize troublesome corrections for air buoyancy and moisture adhering to the surface. Even then a small buoyancy correction is required because the volumes of the evacuated bulbs differ somewhat from that when filled with water. This correction is, however, very small.